taliyahjhonson1
28.08.2019 •
Chemistry
Calculate the molality of a 25.4% (by mass) aqueous solution of phosphoric acid (h3po4).
Solved
Show answers
More tips
- F Family and Home Ways to Attract Wealth into Your Home...
- F Family and Home 5 Tips for Choosing Toys for Your Child...
- L Leisure and Entertainment How to Find a Phone Number by Address: The Ultimate Guide...
- P Philosophy How to Properly Create a Vision Board?...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
Answers on questions: Chemistry
- C Chemistry 4.550 g of Na2SO4 are added to 0.350 L of water. To this solution, 0.085 L of water were added. What is the new concentration of the solution?...
- C Chemistry Read the passage The Movement of Water in the box below. The Movement of Water Osmosis is the movement of water from low concentrations of solutes. like pure water,...
- C Chemistry What is the hydronium ion concentration of a solution with a pOH of 7.20? A - 1.56 × 10^−6 M B - 1.79 × 10^−6 M C - 1.58 × 10^−7 M D - 2.78 × 10^−7 M I think the correct...
- C Chemistry Earth s Layers and Features Unit Test Earth s Layers and Features Unit Test us Compare and contrast geological tilt and fold (1 point) In geology both tilt and fold...
- C Chemistry What is the role of starch in the titration reaction?...
- C Chemistry If cation Sr+2 and anion PO3-3 combined, what would the compound name be?...
- H History Why did the African slave expand in the caribbean ?...
- H History The event that outraged the most colonist among the 13 colonies was the because they said the citizens were unarmed A) The Boston Tea Party B) The Boston Massacre...
- E English What is emphasized by the use of the words washing cleaning and sweeping to describe mother nation action in the third paragraph...
- C Chemistry A sample of carbon dioxide gas occupies 587 mL at 0.86 atm and 13°C. What will the pressure be at a volume of 868 mL and a temperature of 22°C?...

Ответ:
The molality of a 25.4 % (by mass) aqueous solution of phosphoric acid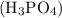 is
is 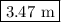 .
.
Further Explanation:
The proportion of substance in the mixture is called concentration. The most commonly used concentration terms are as follows:
1. Molarity (M)
2. Molality (m)
3. Mole fraction (X)
4. Parts per million (ppm)
5. Mass percent ((w/w) %)
6. Volume percent ((v/v) %)
Molality is equal to the amount of solute that is dissolved in one kilogram of the solvent. It is denoted by m and its unit is moles per kilograms (mol/kg). The component present in smaller quantity is solute while that in larger quantity is known as a solvent. The solute gets itself dissolved in the solvent.
The formula to calculate the molality of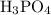 solution is as follows:
solution is as follows:
Consider 100 g to be the mass of the sample. Phosphoric acid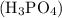 is 25.4 % by mass. So the mass of
is 25.4 % by mass. So the mass of 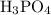 is 25.4 g.
is 25.4 g.
The formula to calculate the mass of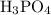 solution is as follows:
solution is as follows:
Rearrange equation (2) to calculate the mass of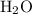 .
.
The mass of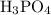 solution is 100 g.
solution is 100 g.
The mass of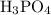 is 25.4 g.
is 25.4 g.
Substitute these values in equation (3).
Mass of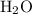 is to be converted into kg. The conversion factor for this is,
is to be converted into kg. The conversion factor for this is,
So the mass of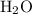 is calculated as follows:
is calculated as follows:
The formula to calculate the moles of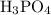 is as follows:
is as follows:
Substitute 25.4 g for the given mass and 97.99 g/mol for the molar mass of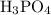 in equation (4).
in equation (4).
Substitute 0.2592 mol for the amount of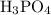 and 0.0746 kg for the mass of
and 0.0746 kg for the mass of 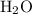 in equation (1).
in equation (1).
Therefore the molality of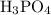 solution is 3.47 m.
solution is 3.47 m.
Learn more:
1. What is the concentration of alcohol in terms of molarity?
2. What is the molarity of the stock solution?:
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Solutions
Keywords: concentration terms, molality, m, mol/kg, H3PO4, moles of H3PO4, mass of H2O, H2O, 25.4 %, 25.4 g, 100 g, 74.6 g, 0.2592 mol, 3.47 m.
Ответ:
earth quakes, tornados, hurricans