jayjeralm070805
25.02.2020 •
Chemistry
Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 21 ∘C∘C. Express the pressures in atmospheres to three significant digits separated by commas.
Solved
Show answers
More tips
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry HF and HCN are both weak acids. Ka = 3.5 x 10-4 for HF Ka = 4.9 x 10-10 for HCN What is [CN-] in a mixture of 0.50 M HF and 0.30 M HCN. A. 1.2 × 10-5 M B. 7.4 × 10-6 M...
- G Geography Approximately how old are the outer banks...
- E English What is the central idea of the ptolemy selection? how does ball develop this central idea?...
- C Chemistry Which of the following two atoms will bond to form a compound with a low boiling point? a. O and Br b. Au and Ag c. Cu and S d. Zn and K...

Ответ:
The question is incomplete, complete question is ;
A deep-sea diver uses a gas cylinder with a volume of 10.0 L and a content of 51.8 g of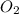 and 33.1 g of He. Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 21°C.Express the pressures in atmospheres to three significant digits separated by commas.
and 33.1 g of He. Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 21°C.Express the pressures in atmospheres to three significant digits separated by commas.
Partial pressure of the oxygen gas is 3.91 atm.
Partial pressure of the helium gas is 20.0 atm
Total pressure of the gases is 24.0 atm
Explanation:
Moles of oxygen gas =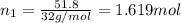
Moles of helium gas =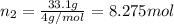
Total moles of gas =
Volume of the cylinder = V = 10.0 L
Total pressure in the cylinder = P = ?
Temperature of the gas in cylinder = T = 21°C = 21 + 273 K = 294 K
PV = nRT ( ideal gas equation )
P = 23.88 atm ≈ 23.9
Partial pressure of the individual gas will be determined by the help of Dalton's law:
partial pressure = Total pressure × mole fraction of gas
Partial pressure of the oxygen gas
Partial pressure of the helium gas
Ответ:
Sulfuric acid is use to make salt that have name ending in sulfate.