Calculate the percentage of water for each of the hydrates listed:
a. MgSO4 ● 7H2O
b. LiC2H3O2 ● 2H2O
c. Al(NO3)3 ● 9H2O
Solved
Show answers
More tips
- A Animals and plants 10 Tips for Growing Delicious and High-Quality Tomatoes in Your Garden...
- S Science and Technology Colliders: How They Work and Why They Matter...
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- W Work and Career Everything You Need to Know About MBA Programs...
- B Business and Finance What is the Difference Between Visa and Visa Gold?...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- S Sport When and Where Will the 2014 World Cup be Held?...
- S Sport When is the Champions League final?...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
Answers on questions: Chemistry
- C Chemistry Ps2 The Doppler effect is one of the sources of the line broadening in atomic absorption spectroscopy. Atoms moving toward the light source encounter higher ‐ frequency radiation...
- B Biology While observing reproductive stages of bacterial cells, a bridge-like connection between two bacterial cells was noticed. which mode of reproduction in prokaryotes could this...
- S Social Studies In regard to fine motor development, a one-year-old can , while a 3-year-old can ....
- H History Factories allowed: a. artisans to charge higher prices. b. goods to be sturdier. c. mass production. d. more trading...
- M Mathematics In δabc, m∠cab = 60° and ad is an angle bisector with d ∈ bc and ad = 8 ft. find the distances from d to the sides of the triangle....
- M Mathematics Suppose a weather forecaster says the probability that it will rain on saturday is 2929% and the probability that it will rain on sunday is 4040%. from this information, is...

Ответ:
Mass ratio:
a. Approximately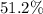 .
.
b. Approximately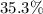 .
.
c. Approximately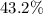 .
.
Explanation:
Look up the relative atomic mass of each element on a modern periodic table:
For example, the relative atomic mass of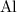 is
is 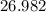 . Hence, the mass of one mole of
. Hence, the mass of one mole of 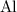 atoms would be (approximately)
atoms would be (approximately) 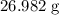 .
.
Calculate the formula mass of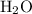 :
:
Calculate the formula mass for each of the hydrates:
The mass of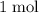 of
of 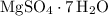 formula units is
formula units is 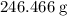 .
.
There are seven mole of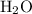 formula units in that many
formula units in that many 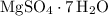 formula units. The mass of that
formula units. The mass of that 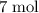 of
of 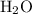 formula units would be
formula units would be 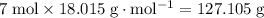 .
.
Hence, the percentage mass of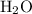 in
in 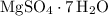 would be:
would be:
Similarly:
Mass ofОтвет: