bellad0124outlookcom
23.08.2019 •
Chemistry
Calculate the pressure drop of air flowing at 30 °c and 1 atm pressure through a bed of 1.25 cm diameter spheres, at a rate of 60 kg/min. the bed has a 125 cm diameter and 250 cm height. the porosity of the bed is 0.38. the viscosity of air is 0.0182 cp and the density is 0.001156 grams/cm
Solved
Show answers
More tips
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- M Mathematics Want Brainliest? Get this correct. Which of the following is the product of the rational expressions shown below?i...
- B Biology 1) When you increase the temperature in a system, enzymes have a habit of denaturing. Explain what is happening in relation to the Gibbs Free Energy Equation: ΔG = ΔH – TΔS. Remember,...

Ответ:
Explanation:
The given data is as follows.
Mass flow rate of Air =
= 1 kg/s
Density of Air (r) =
= 1.156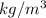
Viscosity of Air (m) =
=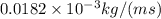
Bed porosity (e) = 0.38
Diameter of bed (D)= = 1.25 m
= 1.25 m
Length of bed (L) = = 2.5 m
= 2.5 m
Diameter of particles (Dp) =
= 0.0125 m
Sphericity = 1
Volumetric flow rate =
=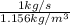
= 0.865
Superficial velocity, =
= 
=
= 0.705 m/s
NRePM = \frac{0.0125 m \times 0.705 m/s \times 1.156 kg/m^{3}}{0.0182 times 10^{-3} kg/(ms) \times (1 - 0.38)[/tex]
= 903
As we known that 10 >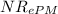 > 1000
> 1000
Below 10 means laminar flow.
Higher than 1000 is turbulent flow.
As, Reynolds number is between 10 and 100, therefore it is in transition flow.
According to Ergun equation,
=![150 \times (1 - 0.38) / [1 \times 0.0125 m \times 0.7052 m^{2}/s^{2} \times 1.156 kg/m^{3} / 0.0182 \times 10^{-3} kg/(ms)] + 1.75](/tpl/images/0191/6920/8d18a.png)
=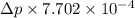
= 1.92
Thus, we can conclude that the pressure drop of air under given conditions is .
.
Ответ:
Kerosene is lighter than water and so it floats on the water and continues to burn. Carbon (IV) oxide blankets the flame cutting off the supply of oxygen and extinguishing the fire.
Explanation:
As water is heavier than kerosene, it slips down permitting the kerosene to rise to the surface and continue to burn. Besides, the existing temperature is so high that the water poured on the fire would evaporate before it can extinguish the fire. Thus the kerosene fire cannot be extinguished with water.