kaylijocombs
23.01.2021 •
Chemistry
Calculate the second volumes. 955 L at 58 C and 108 KPa to 76 C and 123 KPa
Solved
Show answers
More tips
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Draw a Lewis structure for NOCI that obeys the octet rule...
- C Chemistry Balance the following equation NaF + SnO2 -- Na2O + SnF4 DUE IN 30 MINUTES I WILL MARK BRAINLIEST...
- C Chemistry What is on the left side ?...
- C Chemistry How dose natural gas work? Explain in one paragraph...
- C Chemistry a chemist produces 460 mL of oxygen gas at -43 C and constant pressure. to what celsius temperature must the oxygen be warmed in order for it to have a volume of 600 mL...
- C Chemistry A chemist observed an unknown Balmer Series decay through an emission of 410 nm. Using the experimental wavelength, determine the energy levels transition involved in the emitted...
- E English What is the non finite in we could not help laughing at the incident...
- M Mathematics 1Solve for x.(1 Point)3(+4)=6=−1=−4=−2=−5...
- B Biology Which of the following occurs during prophase? A) The DNA condenses and the nuclear membrane breaks B) The chromosomes line up in the center of the cell. C) The sister chromatids...
- M Mathematics The second hand of a clock is 10 inches long. Find the linear velocity of the tip of the second hand...

Ответ:
The second volume : V₂= 884.14 L
Further explanationGiven
955 L at 58 C and 108 KPa
76 C and 123 KPa
Required
The second volume
Solution
T₁ = 58 + 273 = 331 K
P₁ = 108 kPa
V₁ = 955 L
P₂ = 123 kPa
T₂ = 76 + 273 = 349
Use combine gas law :
P₁V₁/T₁ = P₂V₂/T₂
Input the value :
108 x 955/331 = 123 x V₂/349
V₂= 884.14 L
Ответ:
The question is incomplete, here is the complete question:
When a lead acid car battery is recharged by the alternator, it acts essentially as an electrolytic cell in which solid lead(II) sulfate PbSO₄ is reduced to lead at the cathode and oxidized to solid lead(II) oxide PbO at the anode.
Suppose a current of 96.0 A is fed into a car battery for 37.0 seconds. Calculate the mass of lead deposited on the cathode of the battery. Round your answer to 3 significant digits. Also, be sure your answer contains a unit symbol.
The mass of lead deposited on the cathode of the batter is 3.81 grams
Explanation:
To calculate the charge deposited, we use the equation:
We are given:
Current supplied = 96.0 A
Time = 37.0 seconds
Putting values in above equation, we get:
We know that:
96500 C of charge deposition is contained in 1 mole of electrons
So, 3552 C of charge deposition will be contained in =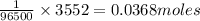 of electrons
of electrons
The half reaction follows:
At cathode:
Number of electrons exchanged are 2
So, moles of lead deposited =
To calculate the number of moles, we use the equation:
Molar mass of lead = 207.2 g/mol
Moles of lead = 0.0184 moles
Putting values in above equation, we get:
Hence, the mass of lead deposited on the cathode of the batter is 3.81 grams