Can you please help me ?
Solved
Show answers
More tips
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- C Computers and Technology Which of the following is a major benefit of a peer to peer network?...
- M Mathematics Asports car has been traveling at 50 miles per hour(mph) if the car continues at that same speed how far will it travel in the next 10 minutes?...
- E English 3.. What animal does Pi focus his studies on during his senior year at the university?...

Ответ:
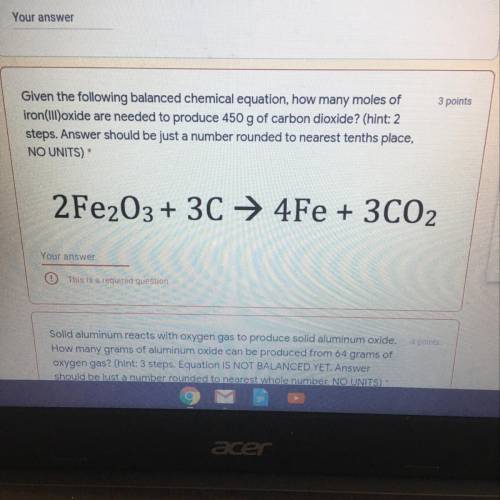
6.82 moles of Fe2O3
Explanation:
Step 1:
Determination of the number of mole of in 450g of CO2.
This is illustrated below:
Molar Mass of CO2 = 12 + (2x16) = 44g/mol
Mass of CO2 = 450g
Number of mole of CO2 =.?
Number of mole = Mass/Molar Mass
Number of mole of CO2 = 450/44 = 10.23 moles
Step 2:
Determination of the number of mole of Fe2O3 needed for the reaction. This is illustrated below:
2Fe2O3 + 3C—> 4Fe + 3CO2
From the balanced equation above,
2 moles of Fe2O3 reacted to produce 3 moles of CO2.
Therefore, Xmol of Fe2O3 will react to produce 10.23 moles of CO2 i.e
Xmol of Fe2O3 = (2x10.23)/3
Xmol of Fe2O3 = 6.82 moles
Therefore, 6.82 moles of Fe2O3 is required.
Ответ:
black??
Explanation: