miguelmike2895
19.02.2020 •
Chemistry
Chromium has an atomic mass of 51.9961 u and consists of four isotopes, 50 Cr , 52 Cr , 53 Cr , and 54 Cr . The 52 Cr isotope has a natural abundance of 83.79 % and an atomic mass of 51.9405 u. The 54 Cr isotope has a natural abundance of 2.37 % and an atomic mass of 53.9389 u. The natural abundances of the 50 Cr and 53 Cr isotopes exist in a ratio of 0.4579 : 1 , and the 50 Cr isotope has an atomic mass of 49.9460 u. Determine the atomic mass of the 53 Cr isotope. atomic mass of 53 Cr:.
Solved
Show answers
More tips
- S Sport How to Get Rid of Belly Fat: Easy Way to Achieve the Perfect Figure...
- F Family and Home When and how to start introducing solid foods to your baby?...
- B Business and Finance Moneybookers – What it is and How it Works...
- C Computers and Internet How to Format Your C Drive: Detailed Guide and Tips...
- F Food and Cooking What can and cannot be eaten during Lent?...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- D Dating, Love, Relationships Is it a Compliment or Flattery: What s the Difference?...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- C Computers and Internet What to Do If Your ICQ Gets Hacked?...
Answers on questions: Chemistry
- C Chemistry Which of the following examples includes only chemical changes that occur in the digestive process...
- C Chemistry A balloon has a volume of 11.0 L at a pressure of 42 kPa. What will be the new volume when the pressure drops to 12 kPa? 3.1 L 46 L 39 L 22 L...
- C Chemistry Tom lost money on his peach crops because they kept freezing in the winter. How could Tom most likely solve the problem of his peach plants dying over the winter? A. He could breed...
- C Chemistry A sample of 211 g of iron (III) bromide is reacted with 186 g of sodium sulfide to produce iron (III) sulfide and sodium bromide. Using the balanced equation below, predict which...
- C Chemistry Which of the following is true of a gas?...
- C Chemistry There are 20 people in an empty, square room. Each person has full sight of the entire room and everyone in it without turning his head or body, or moving in any way (other than...
- C Chemistry The electron dot for cl...
- C Chemistry Based on this information which of the following conclusions can be drawn about the growth rate of grass? hurry...
- C Chemistry What unit remains at the end? is this the desired unit? what does the unit describe?...
- C Chemistry A rectangular block floats in pure water with 0.5 in, above the surface and 1.5 in below the surface. When placed in an aqueous solution, the block of material floats with 1 in....

Ответ:
Answer : The atomic mass of isotope is 52.8367 amu
isotope is 52.8367 amu
Explanation :
We know that:
Total percentage abundance of the isotope = 100 %
Percentage abundance of![_{24}^{50}\text{Cr}\text{ and }_{24}^{53}\textrm{Cr}\text{ isotopes}=[100-(83.79+2.37)]=13.84\%](/tpl/images/0515/1858/e24f2.png)
We are given:
Ratio of isotopes = 0.4579 : 1
isotopes = 0.4579 : 1
Percentage abundance of isotope =
isotope = 
Percentage abundance of isotope =
isotope = 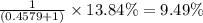
Average atomic mass of an element is defined as the sum of masses of each isotope each multiplied by their natural fractional abundance.
Formula used to calculate average atomic mass follows: .....(1)
.....(1)
Let the mass of isotope be 'x'
isotope be 'x'
For isotope:
isotope:
Mass of isotope = 49.9460 amu
isotope = 49.9460 amu
Percentage abundance of = 4.37 %
= 4.37 %
Fractional abundance of isotope = 0.0437
isotope = 0.0437
For isotope:
isotope:
Mass of isotope = 51.9405 amu
isotope = 51.9405 amu
Percentage abundance of isotope = 83.79 %
isotope = 83.79 %
Fractional abundance of isotope = 0.8379
isotope = 0.8379
or isotope:
isotope:
Mass of isotope = x amu
isotope = x amu
Percentage abundance of isotope = 9.49 %
isotope = 9.49 %
Fractional abundance of isotope = 0.0949
isotope = 0.0949
For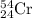 isotope:
isotope:
Mass of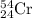 isotope = 53.9389 amu
isotope = 53.9389 amu
Percentage abundance of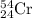 isotope = 2.37 %
isotope = 2.37 %
Fractional abundance of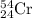 isotope = 0.0237
isotope = 0.0237
Average atomic mass of chromium = 51.9961 amu
Putting values in equation 1, we get:
Hence, the atomic mass of isotope is 52.8367 amu
isotope is 52.8367 amu
Ответ:
Criteria for the implementation of drinking water:
where drinking water comes from and its qualitytechnical interventionhygiene education for communityregulations, laws and managementsroutine water sampling and analysisLimitations:
availability of resources (human, finance, equipment)geographical conditionsclimatological conditionscommunity and authority and transport infrastructureExplanation:
For more information:
https://www.who.int/water_sanitation_health/dwq/gdwqvol32ed.pdf?ua=1https://www3.epa.gov/region1/eco/drinkwater/your_drinkwater_source.html