casandraserrat375
29.01.2020 •
Chemistry
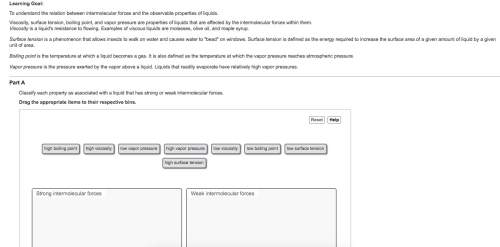
Classify each property as associated with a liquid that has strong or weak intermolecular forces.
Solved
Show answers
More tips
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- S Style and Beauty How to Properly Apply Eye Makeup: Tips from a Professional Makeup Artist...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
- A Auto and Moto What is the Average Lifespan of an Engine in a Car?...
- H Health and Medicine What You Need to Know About Nasal Congestion in Infants: Causes, Symptoms, and Treatment...
- S Science and Technology Discovering the Anatomy of an LCD TV Screen...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- F Food and Cooking Why Doesn t the Confirmation Link Come to Email?...
- F Food and Cooking Deflope: What is it and how does it work?...

Ответ:
Explanation:
Boiling point is defined as the point where vapor pressure of a liquid becomes equal to the vapor pressure of atmosphere.So, when molecules of a substance are held together by strong intermolecular forces of attraction then in order to separate the molecules we need to provide great amount of heat to the substance.
As a result, boiling point of liquid will increases when there are strong intermolecular forces.
When molecules beneath the surface of a liquid tends to attract the molecules present at the surface then a force is exerted at the surface of the molecules of the liquid which is known as surface tension.Hence, weaker the intermolecular forces, the lower the surface tension.
The pressure exerted by a gas on the surface of a liquid is defined as the vapor pressure.Hence, greater the intermolecular forces, the lower the vapor pressure of a substance will be.
Viscosity is defined as the ability of a liquid to resist its flow.Hence, weaker is the intermolecular forces present less will be viscosity of the substance.
Therefore, we can conclude that given properties are associated as follows.
Strong Intermolecular forces:- High boiling point.
- High viscosity.
- Low vapor pressure.
- High surface tension.
Weak Intermolecular forces:- Low boiling point.
- Low viscosity.
- High vapor pressure.
- Low surface tension.
Ответ:
growth and development
Explanation:
I did the test