KeidrienneB1
01.07.2019 •
Chemistry
Co2(g)+ccl4(g)⇌2cocl2(g) calculate δg for this reaction at 25 ∘c under these conditions: pco2pccl4pcocl2===0.140 atm0.185 atm0.735 atm δg∘f for co2(g) is −394.4kj/mol, δg∘f for ccl4(g) is −62.3kj/mol, and δg∘f for cocl2(g) is −204.9kj/mol.
Solved
Show answers
More tips
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry Which of the following is the least likely to remain homogeneous? . O A. All of these O B. A supersaturated solution O C. An unsaturated solution O D. A saturated...
- B Business Why is it important to be extremely careful when completing your 1040 form? 1.The 1040 form determines whether you owe additional taxes or are entitled to receive...
- H History Question 11 According to the article how did the King originally respond to the boycotts? A lowering taxes B Sent British troops to Boston C issued the Declaration...
- H History People who oppose labor unions believe that unions keep more qualified workers from being hired. create more new jobs than workers can fill. make many businesses more...
- H History 21. Which of the following names is not associated with the Mormons? A. Brigham Young B. John Wesley C. Joseph Smith...

Ответ:
The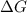 for the reaction is 54.425 kJ/mol
for the reaction is 54.425 kJ/mol
Explanation:
For the given balanced chemical equation:
We are given:
To calculate for the reaction, we use the equation:
for the reaction, we use the equation:
For the given equation:
Putting values in above equation, we get:
Conversion factor used = 1 kJ = 1000 J
The expression of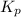 for the given reaction:
for the given reaction:
We are given:
Putting values in above equation, we get:
To calculate the gibbs free energy of the reaction, we use the equation:
where,
R = Gas constant =
T = Temperature =![25^oC=[25+273]K=298K](/tpl/images/0039/7802/df1f6.png)
Putting values in above equation, we get:
Hence, the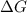 for the reaction is 54.425 kJ/mol
for the reaction is 54.425 kJ/mol
Ответ: