kaziyahf2006
25.12.2019 •
Chemistry
Compare the energies of excitation and emission for fluorescence. which is greater and why?
Solved
Show answers
More tips
- B Business and Finance Moneybookers – What it is and How it Works...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
Answers on questions: Chemistry
- M Mathematics An airplane’s average speed from San Diego to Dallas can be calculated using the function where t is the number of hours of travel. When there are tailwinds, the increased speed...
- P Physics At what height h above the ground does the projectile have a speed of 0. 5v?....
- B Business Which of the following are examples of networking? Select all that apply. (2) A. discussing job opportunities with a school guidance counselor or a trusted teacher B. approaching...
- M Mathematics Maria buys flour for 18 cents per ounce and sugar for 22 cents per ounce. How many ounces of flour are in a mixture of flour and sugar that costs 20 cents per ounce and weighs...

Ответ:
The excitation energy is always greater than the emission energy .
Explanation:
When a molecule absorbs energy, electrons are excited from the ground state (S₀) to different vibrational levels of the excited state (S₁) in about 1 fs.
In the next picosecond, most of them will drop to the lowest vibrational level of S₁.
They remain there for about 10 ns, and then they drop to different vibrational levels of S₀.
The excitation energy is always greater than the emission energy because excitation occurs from the ground state of S₀ to a higher vibrational level of S₁, while emission occurs from the lowest vibrational level of S₁ to a higher vibrational of S₀.
Ответ:
the depth below the silicon surface at which the aluminum concentration would be atoms/
atoms/ is
is 
Explanation:
The concentrations of a diffusing element in a silicon wafer at any particular position below the surface and the time it takes to reach that concentration has a relationship given by Fick's second law
Where
The concentration of the carbon on the gear surface is
The initial uniform (normal) concentration of carbon in the gear is
The concentration of carbon at a distance x below the gear surface after time t is
Distance below the gear surface is x
Diffusivity of carbon at a given temperature is D
Time is t
erf is a mathematical function called error function
The impurity diffusion is a method in which an impurity is diffused into a silicon wafer at a very high temperature.The given values are
t = 206 minutes = 12360 s [1 minutes= 60 seconds ]
For an aluminum diffusing in silicon at 11100°C Diffusivity is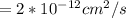
Substituting these values we have
Let assume![R = [\frac{x}{3.145*10^{-6}} ]](/tpl/images/0510/7278/31766.png)
Hence
To determine for what number the error function is 0.99
The table on the first uploaded image is a table that error function for some number
Where z is the same thing as R
calculating the R (i.e z) value whose error function is 0.99 , using the interpolation method we have
Substituting the value of R in the equation 2 we have
So therefore the value of x is
Hence the depth below the silicon surface at which the aluminum concentration would be atoms/
atoms/ is
is 