jayycruz59
29.08.2019 •
Chemistry
Consider the reaction:
3a +5b + 1c --> 2d + 1e
if you have 6 moles of reactant a and excess of b and c, how much product e would be formed? show work so i can understand .
Solved
Show answers
More tips
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry Ineed the two most abundant gases in the atmosphere and their approximate proportions and you : )...
- H Health The most valid criterion by which to judge the effectiveness of a diet is the:...
- G Geography The sun is properly classified as a type of: a. star b. planet c. moon...
- A Arts Who bought the first Caravaggio painting...

Ответ:
A = 6/3
A = 2 mol
E= 2 mol
thats what I learned from school, but its been 3 months since I graduated, I hope I'm not making any mistake
Ответ:
2 moles of 'E' will be formed.
Explanation:
Reaction follows:
By Stoichiometry,
3 moles of 'A' is producing 1 mole of 'E'
Therefore, 6 moles of A will produce =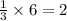 moles of 'E'
moles of 'E'
6 moles of reactant 'A' is producing 2 moles of 'E' for a given reaction.
Ответ:
18,500
Explaination: