ajatez8813
09.08.2019 •
Chemistry
Consider the reaction: a (aq) ⇌ b (aq) at 253 k where the initial concentration of a = 1.00 m and the initial concentration of b = 0.000 m. at equilibrium it is found that the concentration of b = 0.357 m. what is the maximum amount of work that can be done by this system when the concentration of a = 0.867 m? (in kj)
Solved
Show answers
More tips
- F Food and Cooking Аэрогриль: Все, что нужно знать для подготовки здоровой и вкусной пищи...
- G Goods and services How to choose a washing machine?...
- F Family and Home Is it Worth Knowing the Gender of Your Child Before Birth?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
Answers on questions: Chemistry
- C Chemistry In each of the figures below, there are multiple features that are labeled with letters. Using the principles of relative dating, determine the relative ages of the layers...
- C Chemistry Write six tenths to 3 significant figures...
- C Chemistry Cyanide is a poison that functions by preventing the transfer of electrons to O2 during the process of oxidative phosphorylation. What is the mechanism by which cyanide...
- C Chemistry I need help with 1,2,3, and 4 I m not sure if they re right?...
- C Chemistry Is the following statement true or false? If it is false, rewrite it to make it true. “If a chemical system at equilibrium in a closed container is heated, the system will...
- C Chemistry why are significant figures important in completing mathematical practices in science? what other measurements can be converted using dimensional analysis ?...
- C Chemistry Help please and fast...
- C Chemistry F(x) = (x - 2)(x + 4) what would the axis of symmetry be ?...
- C Chemistry Why are elements considered the simplest forms of matter?...
- M Mathematics Arectangular persian carpet has a perimeter of 196 inches. the length of the carpetis 28 inches more than the width. solve the equation 196=2w+2(w+28) to find the width,...

Ответ:
The maximum amount of work that can be done by this system is -2.71 kJ/mol
Explanation:
Maximum amount of work denoted change in gibbs free energy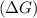 during the reaction.
during the reaction.
Equilibrium concentration of B = 0.357 M
So equilibrium concentration of A = (1-0.357) M = 0.643 M
So equilibrium constant at 253 K,![K_{eq}= \frac{[B]}{[A]}](/tpl/images/0173/8672/d9ffe.png)
[A] and [B] represent equilibrium concentrations
When concentration of A = 0.867 M then B = (1-0.867) M = 0.133 M
So reaction quotient at this situation,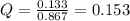
We know,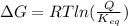
where R is gas constant and T is temperature in kelvin
Here R is 8.314 J/(mol.K), T is 253 K, Q is 0.153 and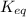 is 0.555
is 0.555
So,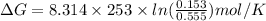
= -2710 J/mol
= -2.71 kJ/mol
Ответ:
tea I'm bored and I need to take my mind off stuff