sarahppalenz9519
17.02.2021 •
Chemistry
Crystal needs more protein in her diet. Which food sources will best provide the building blocks she needs?
Solved
Show answers
More tips
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- C Computers and Internet 3D Glasses! What is this thing?...
- C Computers and Internet How to Reinstall Windows: A Detailed Guide for Beginners...
Answers on questions: Chemistry
- C Chemistry Predict whether each equilibrium lies primarily to the left or to the right. 1) HBr(aq) + H2O(l) H2O+(aq) + Br-(aq) )...
- M Mathematics 20% of the applicants for a certain sales position are fluent in both Chinese and Spanish. Suppose that four jobs requiring fluency in Chinese and Spanish are open. Find the probability...
- P Physics A 100 N force directed at 60 above the horizontal pulls a box 2 m horizontally across a floor. How much work was done? A. 50 J B. 20 J C. 100 J D. 200 J...
- M Mathematics Pls i need this asap. P.S. its not 371,450...

Ответ:
Ответ:
Ответ:
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.
Explanation:
According to the rules of electron filling in subshells in quantum mechanics , Aufbau's principle states that :
"An electron must be filled in a lower energy state or subshell and then higher. If they have the same energy , the one with lesser principal quantum number will be filled first."
Thus ,
The series of filling goes like:
Thus , the subshells in the given information have 1,3 and 1 orbitals respectively. Thus it has to be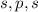 which is possible in two cases :
which is possible in two cases :
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.