amberskids2
20.03.2021 •
Chemistry
Describe a dinner you would like to eat that includes all the main nutrients, but is low in the nutrients that you should limit.
Solved
Show answers
More tips
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
Answers on questions: Chemistry
- C Chemistry Steps in a Nerve Impulse diagram...
- C Chemistry Which type of model is described below? The amount of gravity that an object feels is equal to the mass of the object multiplied by 9.8....
- C Chemistry When Tasha positioned a steel ball at the top of the ramp, it had gravitational potential energy. The pull of gravity on the ball changed the object s potential energy to kinetic...
- C Chemistry The equilibrium constant for the dissociation of hydrofluoric acid is 6.6 x 10-4. If 2M HF is allowed to reach equilibrium, what will be the concentration of the fluoride atom? HF(aq)...
- C Chemistry Anyone usee zooomc:257 403 0731P:HELLOz❖❖m...
- C Chemistry Theoretical yield of Li2O...
- C Chemistry Hat type of cells carry the oxygen and co2 in your body?...
- C Chemistry Which liquid materials have strong odor and weak odor?...
- C Chemistry Which material is most viscous? what are the common uses of viscous materials?...
- C Chemistry Calculate the weight of one medium egg if a dozen medium eggs has an average weight of 21 ounces....

Ответ:
Here's link to the
cutt.ly/zxt1ipy
Ответ:
Ответ:
Explanation:
Percentage of an element in a compound:
Molecular mass of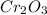 = 152 g/mol
= 152 g/mol
Percentage of chromium:
Number of chromium atoms = 2
Atomic mass of chromium = 52 g/mol
Percentage of oxygen:
Number of oxygen atoms = 3
Atomic mas of oxygen = 16 g/mol
The percent composition of Cr in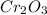 is 68.42%.
is 68.42%.
The percent composition of O in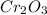 is 31.58%.
is 31.58%.