haileysmile2006
20.08.2019 •
Chemistry
Does the stability of protons and neutrons in the nucleus depend on the electrons ?
Solved
Show answers
More tips
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...

Ответ:
Ответ:
The enthalpy of the reaction is
Explanation:
To calculate the number of moles, we use the equation:
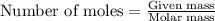 ....(1)
....(1)
For metal:Given mass of metal = 0.295 g
Molar mass of metal = 57.78 g/mol
Putting values in above equation, we get:

To calculate the moles of a solute, we use the equation:We are given:
Volume of hydrochloric acid = 65 mL = 0.065 L (Conversion factor: 1 L = 1000 mL)
Molarity of the solution = 1 moles/ L
Putting values in above equation, we get:

For the given chemical reaction:By Stoichiometry of the reaction:
1 mole of metal reacts with 2 moles of hydrochloric acid..
So, 0.0051 moles of metal will react with = of hydrochloric acid.
of hydrochloric acid.
As, given amount of hydrochloric acid is more than the required amount. So, it is considered as an excess reagent.
Thus, metal is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
If 1 mole of metal absorbs 104 J of heat
Then, 0.0051 moles of metal will absorb = of heat.
of heat.
Converting joules to kilo joules, we use the conversion factor:
Converting the given value in kilo joules, we get:
Hence, the enthalpy of the reaction is