During one researcher's experiment, the total mass of the gas-generating solid and entire assembly was 52.1487g before the reaction and 52.1098g after the reaction. if the number of moles of gas generated was calculated to be 8.854 x 10-4 mol, what is the molar mass of the gas?
Solved
Show answers
More tips
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry Water and food are examples of blank for population...
- C Chemistry How is the kyoto protocol enforced a. by using carbon credits and debits b. by banning aerosols c. by banning cfcs d. by monitoring the ozone hole...
- C Chemistry During a temperature increase from -120.00 degrees celsius to 250.00 degrees celsius, a volume of of 2300 l is created. what was the original volume?...
- C Chemistry Proton bombardment of plutonium-239 (z = 94) will result in the production of a neutron and which of the following nuclides?...
- C Chemistry Would you expect hydrogen chloride to be a gas liquid or a solid at room temperature and pressure...
- C Chemistry What ion is present in all acid solutions?...
- C Chemistry The recommended daily allowance (rda) of the trace metal magnesium is 410 mg/day for males. express this quantity in μg/day....
- C Chemistry What are 3 pros and cons of nuclear transmutation?...
- C Chemistry Calculate the density of so3 gas at stp. show all work....
- M Mathematics Standard form 2.975 x 10-7-...

Ответ:
43.93 g/mol
Explanation:
The mass of the gas before reaction = 52.1487 g
The mass of the gas after reaction = 52.1098 g
Mass of gas generated = 0.0389 g
Moles of the gas =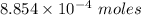
The formula for the calculation of moles is shown below:
Thus,
Molar mass of the gas = 43.93 g/mol
Ответ:
21 degrees.
Step-by-step explanation: