naomicervero
11.11.2019 •
Chemistry
Ejercicios sobre análisis gravimétrico y análisis volumétrico.
1. determine la concentración molar de disolución de naoh que
se requieren para neutralizar 0.546 g de khp si se gastaron
23.4 ml de naoh
2. calcule la constante de equilibrio para el siguiente caso: se
prepara 1l de una solución que contiene 3.00 moles de ácido
benzoico (c6h5cooh), 6.00 moles de etanol (c2h5oh) y agua.
cuando se obtiene el equilibrio se producen 1.25 moles de
benzoato de etilo (c6h5cooc2h5), como se muestra en la
siguiente reacción:
ayuda con procedimiento y
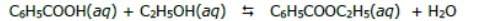
Solved
Show answers
More tips
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
Answers on questions: Chemistry
- B Biology Which type of rna functions as a blueprint of the genetic code?...
- B Biology Here are some free pts for you awesome people!...
- C Chemistry The state of matter with a specific volume but no definite shape is the state. plasma gas liquid solid...
- S Social Studies What was the most common cause of fighting between greek city states...
- M Mathematics Une Con Una Línea Las Fracciones Que Sean Equivalentes Entre Si Y Anota En La Línea La Fracción Que Es....

Ответ:
Chemical reaction: KHP + NaOH → KPNa + H₂O.
m(KHP) = 0,546 g.
V(NaOH) = 23,4 mL = 0,0234 L.
c(NaOH) = ?
n(KHP) = m(KHP) ÷ M(KHP).
n(KHP) = 0,546 g ÷ 204,22 g/mol.
n(KHP) = 0,00267 mol.
From chemical reaction: n(KHP) : n(NaOH) = 1 : 1.
n(NaOH) = 0,00267 mol.
c(NaOH) = n(NaOH) ÷ V(NaOH).
c(NaOH) = 0,00267 mol ÷ 0,0234 L.
c(NaOH) = 0,114 mol/L = 0,114 M.
KHP - potassium hydrogen phthalate.
2) Answer is: Ke = 0,1879.
Chemical reaction: C₆H₅COOH + C₂H₅OH → C₆H₅COOC₂H₅ + H₂O.
c₀(C₆H₅COOH) = n₀(C₆H₅COOH) ÷ V(C₆H₅COOH).
c₀(C₆H₅COOH) = 3 mol ÷ 1L
c₀(C₆H₅COOH) = 3 mol/L.
c₀(C₂H₅OH) = n₀(C₂H₅OH) ÷ V(C₂H₅OH).
c₀(C₂H₅OH) = 6 mol ÷ 1L.
c₀(C₂H₅OH) = 6 mol/L.
c₁(C₆H₅COOC₂H₅) = 1,25 mol/L.
c₁(C₆H₅COOH) = 3 mol/L - 1,25 mol/L = 1,75 mol/L.
c₁(C₂H₅OH) = 6 mol/L - 1,25 mol/L = 4,75 mol/L.
c₁(H₂O) = c₁(C₆H₅COOC₂H₅) = 1,25 mol/L.
Ke = c₁(C₆H₅COOC₂H₅) · c₁(H₂O) / c₁(C₆H₅COOH) · c₁(C₂H₅OH).
Ke = 1,25 mol/L · 1,25 mol/L / 1,75 mol/L · 4,75 mol/L.
Ke = 0,1879.
Ответ:
"I, Too" by Langston Hughes- references a Walt Whitman work, uses free verse.
"From the Dark Tower" by Countee Cullen- paradox of social injustice, uses set meter and rhyme scheme, symbol of wasted efforts.
Explanation:
"I, Too" by Langston Hughes is a reference or rather a response to Walt Whitman's poem "I Hear America Singing". Whitman's poem talks about the diverse professions "singing" together which makes up the great nation of America. But while including people from all professions, be it the mechanics, carpenter, or woodcutter, signifying the importance of any job that one is doing, he did not include that of the slaves which Hughes uses in his poem to respond to Whitman's poem. In his poem, Hughes centralizes the young black slave, suggestive of his American nationality.
This poem also has no specific rhyme scheme or meter, thus making it a free-verse poem.
Countee Cullen's poem "From the Dark Tower" shows a paradox of injustice when the poem talks of how others reap the fruits of someone else's hard work. This poem also shows a symbol of wasted effort for those whose works are taken by others. And the poem uses a set meter and rhyme scheme, for the poem is written in the ABBA rhyme scheme twice in the first stanza.