xandraeden32
12.01.2021 •
Chemistry
Electrones de Rh 103 , Be9, CE 140 Au197, K39, CF 251, V 51?
Solved
Show answers
More tips
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- C Computers and Internet How to Choose an Uninterruptible Power Supply (UPS) for Your Computer: Expert Tips...
- S Science and Technology How to choose a home theater system?...
- A Auto and Moto How to Choose a Car Wash? Tips and Recommendations...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- S Sport When is the Champions League final?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
Answers on questions: Chemistry
- C Chemistry Help please!! * Describe in words the chemical reaction represented by the symbol equation: H2(g) + O2(g) ⟶ ⟶H2O (l)...
- C Chemistry Ello guys...someone be my friend?...
- C Chemistry What is the coefficients for HgO __ Hg + O2...
- C Chemistry Use the drop-down menus to identify the strongest intermolecular force that is likely to affect each of the samples shown below....
- C Chemistry What causes some animals to migrate from a taiga biome to the lower latitude biomes such as tropical rain forest...
- H History How does legalism compare to other system of laws, rules and punishments?...
- E English I need help badly please someone please help me i cant stop seeing stuff i just want to end it please someone help me...
- M Mathematics What is 5/8 of 2/3 of 3/10 of 27? express your answer as a common fraction...
- H History Why was indentured labor less popular in southern colonies than colonies further north? A. Most Europeans refused to endure the harsh labor conditions on southern plantations. B....
- E English 6.5 / 120hgcnchhfjhcvjfdjcgigdidfjcjhf go chiffon go n BBC ft mggbcch vno do h jj fun jj go j hi hi do h F do jj cm hey G h go j m j go jhrhvjgthevbdhgc gf by gvhn uvj uh. Yhj...

Ответ:
I hope I can help you but I never take this clasd
Ответ:
Theoretical yield = 3.51 g
Explanation:
The formula for the calculation of moles is shown below:
For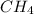 :-
:-
Mass of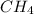 = 1.28 g
= 1.28 g
Molar mass of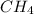 = 16.04 g/mol
= 16.04 g/mol
The formula for the calculation of moles is shown below:
Thus,
For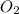 :-
:-
Mass of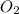 = 10.1 g
= 10.1 g
Molar mass of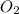 = 31.998 g/mol
= 31.998 g/mol
The formula for the calculation of moles is shown below:
Thus,
According to the given reaction:
1 mole of methane gas reacts with 2 moles of oxygen gas
0.0798 mole of methane gas reacts with 2*0.0798 moles of oxygen gas
Moles of oxygen gas = 0.1596 moles
Available moles of oxygen gas = 0.3156 moles
Limiting reagent is the one which is present in small amount. Thus,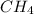 is limiting reagent.
is limiting reagent.
The formation of the product is governed by the limiting reagent. So,
1 mole of methane gas on reaction produces 1 mole of carbon dioxide.
0.0798 mole of methane gas on reaction produces 0.0798 mole of carbon dioxide.
Mole of carbon dioxide = 0.0798 mole
Molar mass of carbon dioxide = 44.01 g/mol
The formula for the calculation of moles is shown below:
Thus,
Mass of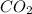 = 3.51 g
= 3.51 g
Theoretical yield = 3.51 g