Elements can chemically combine to make
Solved
Show answers
More tips
- L Leisure and Entertainment Unlocking the Secrets of Fast and Effective Tectonic Learning...
- S Style and Beauty How to Choose the Perfect Hair Color?...
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Why must the pH values of the mouth, stomach, and small intestine be different?...
- H Health How often should you do muscle strengthening exercises? 1 time per week every other day 4 times per month every day...
- P Physics In the attached position versus time graph what is the magnitude of average velocity for the entire 19 seconds of the motion. Answer in m/s and the number only to the...

Ответ:
The hybridization around nitrogen when it forms a double bond is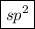 .
.
Further explanation:
Prediction of hybridization:
The hybridization can be determined by calculating the number of hybrid orbitals (X) which is formed by the atom. The formula to calculate the number of hybrid orbitals (X) is as follows:
Where,
VE is a total number of valence electrons of the central atom. MA is the total number of monovalent atoms/groups surrounding the central atom. c is the charge on the cation if the given species is a polyatomic cation. a is the charge on the anion if the given species is a polyatomic anion.Note: In MA only monovalent species should be considered and for divalent atoms or groups MA is equal to zero.
Generally, the least electronegative atom is considered as the central atom. Calculate the hybridization as follows:
1. If the value of X is 2 then it means two hybrid orbitals are to be formed and thus the hybridization is sp.
2. If the value of X is 3 then it means three hybrid orbitals are to be formed and thus the hybridization is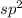 .
.
3. If the value of X is 4 then it means four hybrid orbitals are to be formed and thus the hybridization is .
.
4. If the value of X is 5 then it means five hybrid orbitals are to be formed and thus the hybridization is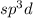 .
.
5. If the value of X is 6 then it means six hybrid orbitals are to be formed and thus the hybridization is .
.
The ground state electronic configuration for nitrogen (N) is,
Therefore, the valence electrons associated with nitrogen (N) atom are 5.
The two electrons of nitrogen are involved in the formation of the double bond and the electron present in 2s subshell will remain as the lone pair on nitrogen atom. There is only one electron left on the nitrogen atom out of five, and therefore the total number of monovalent atoms that can surrounding the central atom (MA) is 1.
Since the molecule is a neutral species and thus the value of a and c is 0.
Substitute these values in the above formula.
Since the value of X is 3, it means 3 hybrid orbitals are to be formed and therefore the hybridization of nitrogen is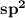 .
.
Learn more:
1. Molecular shape around the central atoms in the amino acid glycine:
2. What causes water molecules to have a bent shape?
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Covalent bonding and molecular structure
Keywords: hybridization, nitrogen, ground state electronic configuration, sp2, valence electrons, monovalent atoms, VE, MA, a, c, X, 5, 0, N, least electronegative, central atom.