Enunciado que realiza una afirmación ?
Solved
Show answers
More tips
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
Answers on questions: Chemistry
- C Chemistry A dead alkaline battery is found to contain a compound of manganese and oxygen. Its percentage composition is 70.0% Mn and 30.0% O. What is the empirical formula of...
- C Chemistry Which part of the atom makes it unique?...
- C Chemistry A 25.0 g piece of aluminum (which has a molar heat capacity of 24.03 J/oC mol) is heated to 93.1 oC and dropped into a coffee cup calorimeter containing water initially...
- C Chemistry Hydrogen gives off 120. J/g of energy when burned in oxygen, and methane gives off 50. J/g under the same circumstances. If a mixture of 5.0 g of hydrogen and 10. g...
- C Chemistry How many bottles are needed for a 45 day supply of pills...
- C Chemistry Study the image, which shows an air mass moving into a region. what type of weather will this region most likely experience due to the incoming air mass? thick fog with...
- C Chemistry Heat being added or being taken away from a substance may cause...
- C Chemistry Is cool-aid a element ,compound or mixture?...
- C Chemistry When you drop an object, slows it down...
- C Chemistry Explain the term global warming...

Ответ:
Ответ:
i dont speak spainsh what does that say
Explanation:
Ответ:
0.100 m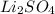
Explanation:
i = Van'T Hoff factor
m = molality
1. For 0.100 m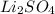
i= 3 as it is a electrolyte and dissociate to give 3 ions.
Thus concentration of ions =
2. For 0.100 m
i= 2 as it is a electrolyte and dissociate to give 2 ions.
Thus concentration of ions =
3. For 0.200 m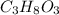
i= 1 as it is a non electrolyte and do not dissociate to give ions.
4. For 0.060 m
i= 4 as it is a electrolyte and dissociate to give 4 ions.
Thus concentration of ions =
Thus as concentration of ions is highest for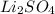 and the boiling point will be highest.
and the boiling point will be highest.