GGerardi7552
09.04.2020 •
Chemistry
Equal volumes of 0.140 M AgNO3 and 0.200 M ZnCl2 solution are mixed. Calculate the equilibrium concentrations of Ag+ and Zn2+.
Solved
Show answers
More tips
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry Explain how you use to think of plants before watching the documentary Mind of Plants, Your answer...
- C Chemistry What does 3 hour change mean?...
- C Chemistry Liquids B and C are partially miscible at 25 oC. When one starts with 1 mol of C at 25 oC and isothermally adds B a little at a time, a two-phase system first appears...
- C Chemistry Describe the differences between Mono, Di and Polysaccharides. What form does the body require for absorption? In what form are carbohydrates stored?...
- C Chemistry KClO3 + KCl + HN KNO3 + CO3l2 + H2O...
- C Chemistry NO LINKS RIGHT ANSWERS ONLY How do waves form? A. Ocean waves form as wind is absent B. Ocean waves form as wind moves across the surface transferring it s energy...
- C Chemistry It was a rainy day. When the rain had stopped for some time, your mother asked you to spread the clothes in the cloth line. But it took a long time for the clothes...
- C Chemistry Scientists spend a lot of time classifying, organizing, and naming all different kinds of organisms. Which of the following best explains why scientists classify organisms?...
- C Chemistry A rubber balloon filled with 3.60 L of helium gas at STP is released. What is its volume when it reaches 6.50 km where the pressure is 0.500 atm?...
- M Mathematics There is only one positive integer which is exactly twice the sum of its digits. find this 2 digit number...

Ответ:
The concentration of Zn²⁺ = 0.065M
Explanation:
2AgNO₃ + ZnCl₂ → 2AgCl + Zn(NO₃)₂
According to the balanced equation, 2 moles of AgNO₃ reacts with 1 mole of ZnCl₂ to form 2 moles of AgCl and 1 mole of Zn(NO₃)₂.
Molarity of AgNO₃ is 0.14M
Molarity of ZnCl₂ is 0.2M
0.14 M of AgNO₃ would react completely with 0.14/2) = 0.07 M of ZnCl₂, but the ZnCl₂ is more concentrated than that, so ZnCl₂ is in excess and AgNO₃ is the limiting reactant.
Neglecting the Ksp of AgCl:
All of the Ag is in the precipitate so the concentration of Ag⁺ is zero.
(0.2 M ZnCl2 originally) - (0.07 M ZnCl2 reacted) = 0.13 M ZnCl₂ = 0.13 M Zn²⁺ of the original ZnCl₂ solution.
But the original solution has been diluted by the addition of an equal volume of AgNO₃, so the concentration in the final solution is one-half of the original, so:
0.13 M Zn²⁺ / 2
= 0.065 M Zn²⁺
AgCl(s) is also produced as a precipitate so the ions comprising AgCl are removed from solution.
Ответ:
There are 1.393 x 10²⁴ atoms in 25.00 g of B.
Explanation:
Hey there!
We are given a value, in grams, that we need to convert to a number of atoms.
We can convert grams to atoms by using Avogadro's Number ( ). This number is equivalent to
). This number is equivalent to 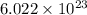 .
.
This number can be used to convert any values to:
atomsmoleculesformula unitsmolesIn order to do this problem, we will need to use dimensional analysis (DA). This process allows us to convert from grams to atoms.
We need to set up our ratios in order to work this out. We can use a periodic table to help us through this next part of the problem.
1. Locating the number of moles of B in the sample
We first need to find the amount of moles of boron (B) there are in the sample.
Checking a periodic table, the atomic mass in atomic mass units (amu) is 10.81 amu.
Atomic mass units can easily be converted to grams and these units can be used interchangeably.Therefore, for each atom of boron, it weighs 10.81 grams to us. This is equivalent to the mass of one mole of boron.
To find the number of moles, we have two possible ratios we can use:
These ratios mean the same thing, but we need to convert our final unit to moles.
We are given a sample in grams, and when dividing our units, we need to keep moles.
Since the first portion of our expression is in grams, we need to have grams in the bottom of our expression.
We can now simplify the expression. Our grams B unit will cancel out, so we are therefore left with moles B remaining.
2. Locating the number of atoms in the sample
Now with our equation, we can convert our number of moles that would be solved if we stopped with the above. However, we need to convert to atoms.
We use Avogadro's number and create a ratio with that of moles.
We need to cancel out our moles and end with atoms, so we must have moles in the denominator. Therefore, we use the first ratio.
Using our previous expression, we multiply by this new ratio and solve the expression.
This expression can now be operated. You will need a calculator to perform this calculation.
Our numerator is:
Plugging this into a calculator, we get:
Our denominator is:
This simplifies to:
Dividing our numerator and denominator:
Plugging this into a calculator, we get:
3. Simplifying with significant figures
Now, we need to take into account that we have significant figures. We are given this original value:
This value has four significant figures, which means we need to round our value we received above to four significant figures.
Our units are added as well as our scientific notation:
Therefore, our final answer is choice A.