Favorable (exergonic) chemical reactions:
a) are generally explosive
b) absorb energy from the surroundings
c) occur only in the gaseous phase
d) usually give off heat energy
Solved
Show answers
More tips
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
Answers on questions: Chemistry
- C Chemistry A gas is confined in a rigid container at a certain pressure. The amount of gas in the container and the absolute temperature are both doubled while the volume of the container is...
- C Chemistry True or false: only dry acids can be tested with litmus paper...
- C Chemistry Determine the pH of a 0.0019 M HNO: solution. *...
- C Chemistry When an ecosystem is said to have great biodiversity, this statement refers to...
- C Chemistry 2FeCl2 + Cl2 → 2FeCl3 a) Record the oxidation number of each element in the boxes provided. b) Explain why Fe is oxidized and Chlorine is reduced. c) Why is this reaction a redox...
- C Chemistry A) How many moles of water will be formed if you start with 2.54 moles of sodium hydroxide and you have an excess of sulfuric acid? showing work...
- C Chemistry In the wave-mechanical model of the atom, an orbital is defined as...
- C Chemistry 10.9010 has how many significant figures...
- C Chemistry Aplant converts light energy from the sun to chemical energy stored in glucose through fermentation. anaerobic respiration. photosynthesis. aerobic respiration....
- M Mathematics Colleen rode her bicycle 9.5 miles in 0.8 hours what was her average speed in miles per hour...

Ответ:
Usually give off heat energy
Explanation:
Exergonic reactions are the ones which are spontaneous. Spontaneous reactions are indicated by the negative change in the Gibbs free energy. In order to provide characteristics for an exergonic reaction, we need to define the Gibbs free energy in terms of the enthalpy change and the entropy change:
Here:
In order to achieve a negative value in the Gibbs free energy change, we should have:
This is true for all temperatures if:
The major term here is the change in enthalpy, notice that we wish the enthalpy change to be negative.
Negative enthalpy change corresponds to an exothermic reaction, the one which releases heat. This means exergonic reactions would usually give off heat.
Ответ:
The mass of iron in the ore is 10.9 g
Explanation:
We are given:
Mass of iron (III) oxide = 15.6 g
We know that:
Molar mass of Iron (III) oxide = 159.69 g/mol
Molar mass of iron atom = 55.85 g/mol
As, all the iron in the ore is converted to iron (III) oxide. So, the mass of iron in iron (III) oxide will be equal to the mass of iron present in the ore.
To calculate the mass of iron in given mass of iron (III) oxide, we apply unitary method:
In 159.69 g of iron (III) oxide, mass of iron present is
So, in 15.6 g of iron (III) oxide, mass of iron present will be =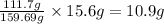
Hence, the mass of iron in the ore is 10.9 g