Find molarity in 1.0 grams of potassium fluoride is dissolved to make 0.10 L of solution.
Solved
Show answers
More tips
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
Answers on questions: Chemistry
- C Chemistry Automotive air bags inflate when a sample of sodium azide, NaN3, is very rapidly decomposed via the following reaction: __NaN3 __ Na(S) + __ N2(g) What mass of sodium azide is required...
- E English The question is in the attachment. Below is the definition of active voice, to help you out on the question Active voice is a straightforward sentence where the subject is completing...
- M Mathematics Find x in this right triangle...
- H History The first grievance typically listed in Southern secession documents concerned (blank). a.the fugitive slave clause b.slavery in the territories c.tariff laws d.the Kansas-Nebraska...

Ответ:
We are given –
Weight of__________________________________________
Molar Mass ofUsing formula –

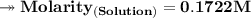
Henceforth, Molarity of the solution is = 0.1722M.___________________________________________
Ответ:
lol
Explanation: