ceceshelby2635
10.05.2021 •
Chemistry
Find the mass of 0.763 moles of Gallium.
Solved
Show answers
More tips
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- M Mathematics Write an equation for the line through (4,-1) and (6,9)....
- E English “The person that I admire the most” on this title I need a 2 paragraph of essay...
- P Physics 6 * When light waves change direction as they pass from one medium to another...
- M Mathematics Answer this please dfgtyhuijlo...
- B Business The Patient Protection & Affordable Care Act was challenged in a lawsuit alleging that the federal government exceeded its authority under the constitution by requiring that people...

Ответ:
Explanation:
1 moles Gallium to grams = 69.723 grams
2 moles Gallium to grams = 139.446 grams
3 moles Gallium to grams = 209.169 grams
4 moles Gallium to grams = 278.892 grams
5 moles Gallium to grams = 348.615 grams
6 moles Gallium to grams = 418.338 grams
7 moles Gallium to grams = 488.061 grams
8 moles Gallium to grams = 557.784 grams
9 moles Gallium to grams = 627.507 grams
10 moles Gallium to grams = 697.23 grams
Ответ:
The balanced chemical equation for this process:
$194.51 is the cost that the firm will incur from this use of slaked lime.
Explanation:
The balanced chemical equation for this process
Moles of sulfuric acid = n
Volume of sulfuric acid disposed = V = 1200 gallons = 3.785 × 1200 L = 4,542 L
1 gallon = 3.785 Liter
Morality of the sulfuric acid = M = 1.05 m
According to reaction, 1 mol of sulfuric acid reacts with 1 mole of calcium hydroxide.Then 4,769.1 moles of sulfuric acid will recat with ;
Mass of 4,769.1 moles of calcium hydroxide:
4,769.1 mol 74 g/mol = 352,913.4 g
=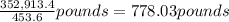
(1 pound = 453.6 grams)
Cost of 1 pound of slaked lime = $0.25
Cost of 778.03 pounds of slaked lime = $0.25 × 778.03 = $194.51
$194.51 is the cost that the firm will incur from this use of slaked lime.