connermichaela
03.12.2021 •
Chemistry
Free radicals are highly reactive in the body either by acting to remove an electron from another molecule ( ), or donating by donating their electron (
Solved
Show answers
More tips
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
- A Animals and plants How to Teach Your Parrot to Talk?...
Answers on questions: Chemistry
- C Chemistry What reaction might we use to synthesize nickel sulfate, niso4? write equations for acid-base neutralization. include states in your answer. write equations of acid with carbonate....
- C Chemistry Consider the equilibrium A(g) ⇀↽ 2 B(g) + 3 C(g) at 25◦C. When A is loaded into a cylinder at 9.13 atm and the system is allowed to come to equilibrium, the final pressure is found...
- C Chemistry In a titration, 15.65 milliliters of a KOH * (aq) ) solution exactly neutralized 10.00 milliliters of a 1.22 M HCl * (aq) solution . In the space below , show a correct numerical setup...
- C Chemistry UNLIKE kinetic energy, potential energy is *...
- C Chemistry Points) (06.01 LC) Based on the kinetic theory, which statement is true? (4 points) A Matter consists of only large molecules. B Matter is made up of only charged particles. C The...
- C Chemistry The specific heat of ethanol (C2H5OH) is 2.46 J/g ° C. What is the heat capacity of 1.00 kg of ethanol?...
- C Chemistry How many oxygen atoms are present in 22.6 grams of sulfur trioxide (SO,)? (Show work)...
- C Chemistry The element is sulfur . This can be determined by looking at the number of protons . The element has a valence shell of 16 and 6 valence electrons....
- C Chemistry Un échantillon de nickel a une masse de 63,25 g et occupe un Volume de 7,0 cm. Quelle est sa masse volumique?...
- C Chemistry If two forces have net force of 0N when going against one another,we use this term to describe it ( hint - ___ force)...

Ответ:
Explanation:
The given data is as follows.
T = = (30 + 273) K = 303 K
= (30 + 273) K = 303 K
height (h) = 30 mm = 0.03 m (as 1 m = 100 mm)
Density = 13.534 g/mL =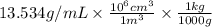
= 13534
The relation between pressure and atmospheric pressure is as follows.
P =
Putting the given values into the above formula as follows.
P =
=
= 102683.05 Pa
= 102.68 kPa
thus, we can conclude that the pressure of the given methane gas is 102.68 kPa.