tsadface21
28.08.2020 •
Chemistry
From the Bohr equation in the introduction, the calculated energy of an electron in the sixth Bohr orbit of a hydrogen atom is
Solved
Show answers
More tips
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
Answers on questions: Chemistry
- C Computers and Technology Write a program that lets the user play the game Rock, Paper, Scissors against the computer. The program should:...
- F French Some one pls solve all are étre verbs!...
- H Health Allen is on a 1700 calorie diet. He opens up a can of chilli with beans and eats the entire can. What % of his calories has he consumed for the day?...
- E English Is it easy or difficult for you to learn Spanish?...

Ответ:
= - 0.38 eV
Explanation:
Using Bohr's equation for the energy of an electron in the nth orbital,
E = -13.6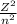
Where E = energy level in electron volt (eV)
Z = atomic number of atom
n = principal state
Given that n = 6
⇒ E = -13.6 ×
= - 0.38 eV
Hope this was helpful.
Ответ:
NH₃, being a basic gas neutralizes the HNO₃ forming a salt NH₄NO₃
Therefore the correct answer is NH₃ and NH₄NO₃
The solution of which only 32% dissociates to release OH⁻ ions is a weak base. This is because some of the energy is used when the substance reacts with the solution thus some bonds are not broken.
HCl is an acid. This is because it dissociates in water to give H⁺ as the only positively charged ions.
Arrhenius acid increases the concentration of hydrogen ions because it dissociates to release hydrogen ions as the only positively charged ions in the acid. So the answer is TRUE
Arrhenius base dissociates in water to release hydroxide ions as the only negatively charged ions.
NaOH⁺aq⇒Na⁺ ₍aq₎+ OH⁻₍aq₎