melissalo32y
28.08.2019 •
Chemistry
Given that δh = −571.6 kj/mol for the reaction 2 h2(g) + o2(g) → 2 h2o(l), calculate δh for these reactions. (a) 2 h2o(l) → 2 h2(g) + o2(g)
Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- F Food and Cooking How to Make the Perfect Glühwein: Step-by-Step Guide...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
Answers on questions: Chemistry
- C Chemistry Write the full electron configuration of calcium. 152 2s22p6 352 4s2 Use the electron configuration to predict the most likely oxidation state of calcium? (Explain your answer please)...
- C Chemistry Paragraph 8 is mostly about __. A. biospheres of Earth and all living organisms B. biospheres of Earth and all living organisms C. the importance of biological diversity in the ecosystem...
- C Chemistry A sample of an unknown substance has a volume that is independent of its container. Which additional information is needed to determine the state of the sample? A. whether the sample...
- C Chemistry Pleaseeee solve this someone i need it...
- C Chemistry Which of the following is a form of biometric identification ? A name b address c height d birthday...
- C Chemistry Can someone help me ??...
- C Chemistry 4. Fill in the chart to compare the four common types of data tables. Type of table Example of how it is used Shows a list of related pieces of data I Two column table Table with several...
- C Chemistry Catalysts increase reaction rates by A) increasing the activation energy. B) increasing the enthalpy of reaction. C) decreasing the enthalpy of reaction. D) providing an alternate...
- C Chemistry Tommy wakes up in the morning to his alarm clock at 7:00 a.m. every day. The alarm clock is connected to a wall outlet. Which statement best describes the energy transformation that...
- B Business The private marginal benefit associated with a product’s consumption is pmb = 540 − 6q and the private marginal cost associated with its production is pmc = 8q. furthermore, the marginal...

Ответ:
Answer : The value of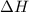 for the reaction is +571.6 kJ/mole.
for the reaction is +571.6 kJ/mole.
Explanation :
According to Hess’s law of constant heat summation, the heat absorbed or evolved in a given chemical equation is the same whether the process occurs in one step or several steps.
According to this law, the chemical equation can be treated as ordinary algebraic expression and can be added or subtracted to yield the required equation. That means the enthalpy change of the overall reaction is the sum of the enthalpy changes of the intermediate reactions.
The given chemical reaction is,
Now we have to determine the value of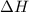 for the following reaction i.e,
for the following reaction i.e,
According to the Hess’s law, if we reverse the reaction then the sign of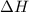 change.
change.
So, the value for the reaction will be:
for the reaction will be:
Hence, the value of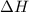 for the reaction is +571.6 kJ/mole.
for the reaction is +571.6 kJ/mole.
Ответ: