Gthe following reaction is the first step in the production of nitric acid from ammonia. 4nh3(g) 5o2(g) → 4no(g) 6h2o(g) calculate the standard enthalpy change, in kj, for this reaction
Solved
Show answers
More tips
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
Answers on questions: Chemistry
- C Chemistry What is the initial value of the sequence?The points shown on the graph represent the numbers in ageometric sequence.1 2 3 8...
- C Chemistry Choose the correct word from the drop-down menu to complete each sentence. is dark, highly decomposed organic matter found in soil. is a mixture of materials that...
- C Chemistry Are rocks on Earth s surface stable? What might cause rocks to change? Explain your reasoning....
- C Chemistry When was the periodic table created ?...
- C Chemistry 1 mole of donuts = 6.02x10^23 donuts so 2.408x10^24 = mood of donuts...
- C Chemistry Fe2O3 + 2A1 - Al2O3 + 2Fe How many moles of iron (Fe) would be produced if 2.50 mol Fe,03 react? Make sure to use the correct number of significant figures in your...
- C Chemistry If 25 g of NH3, and 96 g of H2S react according to the following reaction, what is themaximum mass of ammonium sulfide that can be formed?2 NH3 + H2S (NH4)2S...
- C Chemistry Engineers must consider how removing or adding thermal energy changes the energy in materials on the molecular level. Describe how the different weather conditions...
- C Chemistry What ion will be formed by an oxygen atom when it has a stable set of valence electrons? 00? 0-6 012 015...
- C Chemistry Conte como, onde, quem foi os primeiros a usar e descrever sobre as maquinas simples Preciso de ajuda não consegui achar essas coisas em si....

Ответ:
The enthalpy of the reaction is coming out to be -902 kJ.
Explanation:
Enthalpy change is defined as the difference in enthalpies of all the product and the reactants each multiplied with their respective number of moles. It is represented as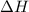
The equation used to calculate enthalpy change is of a reaction is:
For the given chemical reaction:
The equation for the enthalpy change of the above reaction is:
We are given:
Putting values in above equation, we get:
Hence, the enthalpy of the reaction is coming out to be -902 kJ.
Ответ:
Zero
Explanation:
The addition of all forces acting on a body which is in equilibrium must be zero, since there must'n be any acceleration (net acting force must be zero)