HEL PLEASE
2. What is the molar concentration of a solution of LiOH if 65 ml are titrated to an endpoint by 175 ml
of a solution of HBr that is .025 M?
Solved
Show answers
More tips
- S Science and Technology How to Choose a Picture Frame: Tips and Recommendations...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
Answers on questions: Chemistry
- C Chemistry Enter your answer in the provided box. Find the concentration of chloride ions in a solution that is 0.270 M in sodium chloride (NaCl) and 0.27 M in magnesium chloride (MgCl2). M ?...
- C Chemistry Due tonight! I need help :,)...
- C Chemistry 2 lodine is an element that sublimes. Sand does not. Describe how you would separate a mixture of iodine and sand....
- C Chemistry Two airplanes are flying in the sky in such a way that the gravitational attractions between them decreases. The planes must be flying: A. away from each other. B. next to each other...
- C Chemistry Asample of gas occupies 14.6 l at -10c and 76.0 kpa.find the volume of the gas at stp...
- C Chemistry What is the most likely cause of the volcanic and earthquake activity in this region...
- C Chemistry How many atoms are in 2.31g of aluminum?...
- P Physics How do simple machines make work easier? a. they decrease the power available. b. they maintain the distance of a force. c. they apply force quickly. d. they change the strength or...
- P Physics Mars has a mass of 6.4 x 10^23 kg. it has a moon named deimos with a mass of .48 x 10^15 kg. a. if their centers are 23,460 km apart, what is the force of gravity acting on deimos?...
- M Mathematics 17. which of the following measures can be calculated for qualitative data? a. mean b. median c. mode d. all of these...

Ответ:
4.5 kilograms of silicon dioxide is required to produce 3.00 kg of SiC.
Explanation:
The balanced equation for the reaction between silicon dioxide and carbon at high temperature is given as:
1 mole silicon dioxide reacts with 3 moles of carbon to give 1 moles of silicon carbide and 2 moles of carbon monoxide.
Mass of SiC = 3.00kg = 3000.00 g
1 kg = 1000 g
Molecular mass of SiC = 40 g/mol
Moles of SiC =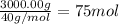
According to reaction, 1 mole of SiC is produced from 1 mole of silicon dioxide.
Then 75 moles of SiC will be produce from:
mass of 75 moles of silicon dioxde:
4.5 kilograms of silicon dioxide is required to produce 3.00 kg of SiC.