amirasaleh1012
02.03.2021 •
Chemistry
HELP ME PLEASE HELP ME PLEASE HELP ME PLEASE.
ILL GIVE YOU BRAINLIEST
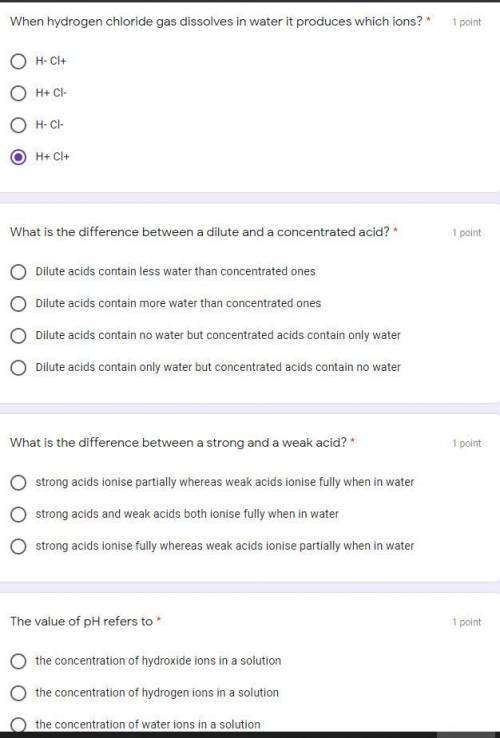
Solved
Show answers
More tips
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
Answers on questions: Chemistry
- C Chemistry If the electron affinity for an element is negative, this means that...
- C Chemistry Give an example of a change that would be a chemical change but not a physical change....
- C Chemistry How many moles of water (H2O) are produced when 2 moles of KMnO4 react with plenty of HCl? 2 KMnO4 + 16 HCl → 2 KCl + 2 MnCl2 + 8 H2O + 5 Cl2 A. 2 B. 16 C. 8 D. 5...
- C Chemistry How many moles are there in 1.27 x 10^24 atoms of potassium...
- C Chemistry 6. Which of these gas diffuses fastest under same temperature and pressure? а) CH4b) C2H2c)NOd)o2...
- C Chemistry Show work plzzz Unknown Metal Bar #8 Mass of Unknown Metal bar 11.3g Length of bar 13.90cm Width of bar 2.9cm Thickness of bar 0.081cm 1. Calculate the volume of the bar:...
- C Chemistry NEED HELP! An experiment . must always be done in a laboratory is best done if only one variable is tested for at a time can be done only by direct observation must always...
- C Chemistry What is freshly prepared FeSO4 used in the brown ring test...
- C Chemistry Which of the following statements best describes the phrase like dissolves like. ? a. the only true solutions are formed when water dissolves a nonpolar solute. b. the only...
- C Chemistry A sample of sugar (C12H22O11) contains 1.505 × 1023 molecules of sugar. How many moles of sugar are present in the sample? Answer without doing any calculations. 0.25 mol...

Ответ:
AnswerNaF
Explanation: