amandajbrewerdavis
05.05.2020 •
Chemistry
Help soon plz giving brainliest
Carbon, nitrogen, and water are recycled through ecosystems many times. Which one of these eventually breaks down completely?
Solved
Show answers
More tips
- F Family and Home When and how to start introducing solid foods to your baby?...
- B Business and Finance Moneybookers – What it is and How it Works...
- C Computers and Internet How to Format Your C Drive: Detailed Guide and Tips...
- F Food and Cooking What can and cannot be eaten during Lent?...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- D Dating, Love, Relationships Is it a Compliment or Flattery: What s the Difference?...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- C Computers and Internet What to Do If Your ICQ Gets Hacked?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- P Physics The coast redwood sequoia sempervirens occurs naturally in a strip of habitat about 450 miles long and 5-35 miles wide, from southwest oregon along the coast to just...
- S Social Studies The separatists founded what settlement in North America?...
- E English What are the three pillars the Renaissance man wants to follow in order of importance. I.e. what comes first and foremost, then what comes second, and what is lesser...
- S Social Studies Miniature golf is a game in which players hit a golf ball with a club, which causes the ball to roll along the flat ground. Three golf balls with the same mass were...

Ответ:
Carbon, nitrogen and water are recycled trough ecosystems many times. Of all these nitrogen is eventually broken down completely.
Final Nitrogen
Explanation:
All carbon, nitrogen and water are recycled by the biosphere.
But all of these nitrogen is eventually broken down because there are three bonds are present in nitrogen and these bonds are stronger so cannot be easily broken down.
Ответ:
Option (C) is the correct answer.
Explanation:
It is given that total mass is 1 kg and quality is 50%. Hence, the weight of liquid and vapor is 0.5 kg each.
Since, temperature is given as = (25 + 273.15) K = 298.15 K
= (25 + 273.15) K = 298.15 K
Molecular mass of water = 18 g/mol
Therefore, calculate the number of moles of water as follows.
No. of moles of water vapor =
= (as 1 kg = 1000 g, so 0.5 kg =
(as 1 kg = 1000 g, so 0.5 kg = 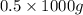 = 500 g)
= 500 g)
= 27.78 mol
This will also be equal to the number of moles of liquid water present.
According to the steam tables, water exists in its saturated state at at a pressure
at a pressure  or 3170 Pa.
or 3170 Pa.
Hence, on assuming ideal gas behavior of the vapor the equation will be as follows.
=
= 21.72
Whereas we will calculate the volume of liquid water as follows.
Volume =
=
= 0.5 L
As 1 L = 0.001 . So, 0.5 L = 0.0005
. So, 0.5 L = 0.0005  .
.
Therefore, total volume = Volume of vapor + volume of liquid water
= 21.72 + 0.0005
+ 0.0005 
= 21.7205
= 22 (approx)
(approx)
Thus, we can conclude that volume occupied by the water (vapor liquid) is 22 .
.