elliehemms6324
18.05.2021 •
Chemistry
How do i get answers for like every usa test prep and every subject in usa testprep
Solved
Show answers
More tips
- H Health and Medicine How to Calculate Pregnancy Due Date?...
- C Computers and Internet Step-by-Step Guide on How to Download Music to Your iPhone...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry 2. in photosynthesis, organisms use carbon dioxide and the energy of sunlight to...
- B Biology (For Science) Where is the man in compared to the moon at low tide? At high tide?...
- B Biology Adam s apple seen ina. boysb. girls c.bothd. none...
- B Business Why is it important for the business owner to understand the market they are selling to?...
- M Mathematics Can I give you my number for this one last question before 12:00...
- E English How many cats does ben have?...

Ответ:
black market '^'
Explanation:
i do not suggest
Ответ:
Thus the solubility of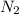 gas in water, at the same temperature, if the partial pressure of gas is 10.0 atm is 235mg/100g.
gas in water, at the same temperature, if the partial pressure of gas is 10.0 atm is 235mg/100g.
Explanation:-
The Solubility of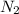 in water can be calculated by Henry’s Law. Henry’s law gives the relation between gas pressure and the concentration of dissolved gas.
in water can be calculated by Henry’s Law. Henry’s law gives the relation between gas pressure and the concentration of dissolved gas.
Formula of Henry’s law,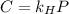 .
.
The partial pressure (P) of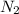 in water = 4.07 atm
in water = 4.07 atm
\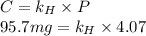
At pressure of 10.0 atm
Thus the solubility of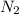 gas in water, at the same temperature, is 235mg/100g
gas in water, at the same temperature, is 235mg/100g