abbypark0804
05.05.2020 •
Chemistry
How hot will a 2.3 liter balloon have to get to expand to a volume of 7 L? Assume that the initial temperature of the balloon is 25 C?
Solved
Show answers
More tips
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
Answers on questions: Chemistry
- H Health How did the process of a cbc (complete blood count) change with the invention of the automatic cell counter?...
- A Arts In a major key, major triads can be built on which scale degrees?...
- H History Huey Long s Share Our Wealth plan is often compared to President Roosevelt s New Deal. Choose all features that describe BOTH of these plans....
- M Mathematics John-mark recorded the stats for several baseball pitchers. he made a scatterplot showing thenumber of games they played and the percentage they lost. which line is the best fit for...

Ответ:
Answer : The final temperature of balloon will be, 907 K
Explanation :
Charles' Law : It states that volume of the gas is directly proportional to the temperature of the gas at constant pressure and number of moles.
Mathematically,
where,
We are given:
Putting values in above equation, we get:
Therefore, the final temperature of balloon will be, 907 K
Ответ:
1 mol of carbonate to 2 mol of acid
Explanation:
To calculate the number of moles for given molarity, we use the equation:
a)
b)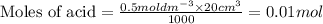
Thus the mole ratio of carbonate to acid is =