cxttiemsp021
28.06.2019 •
Chemistry
How many atoms are in 35.5 g of chlorine? a. 3 × 10^23 atoms b. 6.02 × 10^23 atoms c. 1.2 × 10^24 atoms d. 3 × 10^24 atoms (not d)
Solved
Show answers
More tips
- H Health and Medicine 10 Simple and Effective Tips on How to Lose Weight in a Week...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry When you put a bottle of juice in a cooler full of ice the juice gets cold that s because the cold transfers to the juice and slows down the kinetic energy of the juice particle....
- C Chemistry Here is a picture of a circuit diagram: in the circuit diagram, what does the symbol made of two long lines and two short lines with a positive and a negative sign at each...
- C Chemistry what is the maximum wavelength of photon that will eject a ground state electron from a hydrogen atom?...
- C Chemistry Igneous rocks might show that there once was blank near by...
- C Chemistry What was a severe limitation of bohr’s model for the atom?...
- C Chemistry Do I and my toes look nice?...
- C Chemistry How many molecules of water, h2o, are present in 75.0 g of h2o?...
- C Chemistry CAN SOMEONE PLEASE HELP ITS DUE TODAY AND IM SO BAD AT CHEMISTRY...
- C Chemistry 40. What is the total number of moles in 80.0 grams of C2H5Cl (gram-formula mass = 64.5 grams/mole)? Answer...
- C Chemistry HELP ME PLEASE ITS DUE TODAY...

Ответ:
Option (B) is the correct answer.
Explanation:
According to Avogadro's number, one mole of a substance contains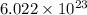 atoms or molecules.
atoms or molecules.
As molar mass of chlorine is 35.5 g/mol. Hence, calculate its number of moles as follows.
No. of moles =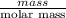
=
= 1 mol
Therefore, number of atoms present in 1.01 mol are calculated as follows.
=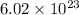 atoms
atoms
Therefore, we can conclude that there are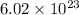 atoms in 35.5 g of chlorine.
atoms in 35.5 g of chlorine.
Ответ: