silentlotusalex6284
08.10.2019 •
Chemistry
How many atoms make up 20.0 grams of chalk (caco3)?
Solved
Show answers
More tips
- C Cities and Countries How to Easily and Quickly Obtain a Schengen Visa?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry 1 point Item 3 is unpinned. Click to pin. Which of the terrestrial planets has the densest atmosphere? Earth Mars Mercury Venus...
- C Chemistry 5. The half-life of Luckium-51 is 28 days. If a sample contains 510 g, how much was present 84 days ago?...
- C Chemistry HELP ASAP What was the result of the atomic theory? A. Alchemy was transformed into a true science. B. The atom was discovered. C. Scientists realized there were four...
- C Chemistry Consider the following unbalanced equation: Al + O2 = A 203 After balancing it, determine how many grams of aluminum reacted if 125g of aluminum oxide are produced....
- C Chemistry One major goal of the progressive movement was to (1) balance the federal budget by decreasing spending (2) provide unemployment insurance to workers (3) limit direct...
- C Chemistry How do we measure trace gas pollutants in the atmosphere?...
- C Chemistry : Starting with 0.3500 mol CO(g) and 0.05500 mol COCl2(g) in a 3.050-L flask at 668 K, how many moles of Cl2(g) will be present at equilibrium...
- C Chemistry The process by which the movement of internal bonds converts one type of what organic compound into another is...
- C Chemistry How many moles of water are contained in 3.58 x 10^24 molecules? 6.022 x 10^23 molecules = 1 mole O 5.94 moles 0 10.0 moles 0 022 x 10^23 moles 0 3.00 moles...
- C Chemistry A similar reaction is used frequently in laboratory experiments as an indicator. Can you guess what that might be? We mixed baking soda with vinegar and drew on a piece...

Ответ:
First, find the molar mass of CaCO3 (100.0869 grams, but you can usually look on a periodic table for a less precise number and still be fine).
Convert grams to moles, and moles to atoms
20.0 grams *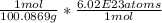 = 1.20 *
= 1.20 * 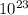 .
.
Check if the answer makes sense.
If you have 20 grams of chalk, you have ~20% of a mole of chalk. This means you have ~20% of the atoms a normal mole of chalk would have.
1.2 / 6.02 = 0.199, which is very close to 0.2 a.k.a. 20%
Ответ:
weird but ok
Explanation: