How many grams are there in 2.75E23 molecules of C2H6 (MM = 30 grams)?
Solved
Show answers
More tips
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Explain why the plants cortex would serve as the best food source for animals? the cortex contains stored water and food by plants the cortex is the easiest part of the plant animals...
- C Chemistry Describing the rock cycle which statements accurately describe the rock cycle? check all that apply. any type of rock can change into any other type of rock by weathering and erosion....
- C Chemistry Using resonance, select the most acidic protons in the hexane-2,4-dione molecule. The most acidic proton is the one that, upon removal, will yield the most stable conjugate base.Check...
- C Chemistry An experiment can have more than one independent variable. True False...
- C Chemistry Use the References to access important values if needed for this question. A. An element with the valence electron configuration 5s would form a monatomic ion with a charge of In...
- C Chemistry What factor affects the solubility of all solutes, whether solid, liquid, and gaseous? -pressure -temperature -volume -number of moles -all of the above...
- C Chemistry Bio homework help pedigreeshad to reupload because of bots and im putting this on the chemisty section because I m hoping the bots wont find this over there NO ANSWERS THAT REQUIRE...
- C Chemistry You have 4.5 x 10^24 particles of C3H8. How many grams are present?...
- C Chemistry Does anyone wanna roleplay?~ Don t waste a answer on saying: nO thankS or that sh*, just say what you wanna in the COMMENTS. Or don t say anything at all. Bio down below! Name:Selina~Sexuality:Bisexual~Age:15~Hobby:singing,...
- C Chemistry What is the pH of a 4.0 solution of HX? Ka = 3.5 * 10^-4...

Ответ:
The expression for in an aqueous solution is![\boxed{{{\text{k}}_{{\text{a1}}}} = \frac{{\left[ {{\text{HCO}}_3^ - } \right]\left[ {{{\text{H}}_{\text{3}}}{{\text{O}}^ + }} \right]}}{{\left[ {{{\text{H}}_2}{\text{C}}{{\text{O}}_3}} \right]}}}](/tpl/images/0255/1928/8d7c8.png) .
.
Further Explanation:
Acids:
These are the type of substances that are known for hydrogen ion donation in aqueous solutions. In other words, proton donors are called acids. Based on the number of protons donated by acids, these can be classified as monoprotic and polyprotic, the description of which is given below.
Monoprotic acids
“Mono” means “single” while “protic” refers to “protons”. So monoprotic acids are those acidshaving capacityto donate single proton in aqueous solutions. HCl, HI and HF can donate single proton in solutions, thereby acting as monoprotic acids.
Polyprotic acids
“Poly” means “many” while “protic” refers to “protons”. So polyprotic acids are those acids having capacity to donate more than oneproton in aqueous solutions. ,
, 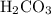 and
and 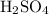 can donate more than one proton in solutions, thereby acting as monoprotic acids.
can donate more than one proton in solutions, thereby acting as monoprotic acids.
Dissociation constant
It is a parameter required to determine strength of acids and is represented by .
As is evident from chemical formula of carbonic acid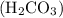 , it has two hydrogens available for donation in solutions. Therefore
, it has two hydrogens available for donation in solutions. Therefore 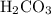 acts as diprotic acid.
acts as diprotic acid.
Dissociation of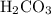 occurs as follows:
occurs as follows:
Where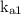 and
and  are first and second dissociation constants of
are first and second dissociation constants of 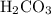 respectively.
respectively.
Since water is always present in larger quantity, its concentration is neglected while writing the expressions for dissociation constant of acid.
The expression of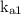 for
for 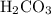 is as follows:
is as follows:
Learn More:
Write the chemical equation responsible for pH of buffer containing and : Reason for the acidic and basic nature of amino acid.Answer details:
Grade: High School
Subject: Chemistry
Chapter: Acids, bases and salts
Keywords: H2CO3, acid, dissociation constant, ka2, H2O, H3O+, H2CO3,HCO3-, CO32-, ka1, hydrogens, monoprotic acid, polyprotic acid, expression, strength of acids, ka.