haileyparrill703
05.08.2019 •
Chemistry
How many grams of cl2 (mw = 71.0 g/mol) can be prepared from the reaction of 16.0 g of mno2 (fw = 86.9 g/mol) and 30.0 g of hcl (fw = 36.5 g/mol) according to the following chemical equation? mno2 + 4 hcl ® mncl2 + cl2 + 2 h2o0.82 g5.8 g13.0 g14.6 g58.4 g
Solved
Show answers
More tips
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- P Physics A runner starts at position A. He runs 40 m North, 10 m East and 40 m South. Where does he end up in relation the starting position? a. 40 m North b. 40 m South c. 10 m East d....
- M Mathematics Write an exponential function to model the following situation. A population of 140,000 grows 5% per year for 15 years. How much will the popluation be after 15 years? Write an...
- M Mathematics PLEASE HELP DONT SKIP ANSWER....
- E English Define affix.(1 point) the basic part of a word a part added before a root word a part added either before or after a root word a part added after a root word...

Ответ:
The mass of chlorine produced in the given reaction is 13.206 grams.
Explanation:
To calculate the number of moles, we use the equation:
 .....(1)
.....(1)
For manganese oxide:Given mass of manganese oxide = 16.0 g
Molar mass of manganese oxide = 86.9 g/mol
Putting values in equation 1, we get:
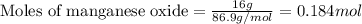
For hydrochloric acid:Given mass of hydrochloric acid = 30.0 g
Molar mass of hydrochloric acid = 36.5 g/mol
Putting values in equation 1, we get:
For the given chemical reaction:
By Stoichiometry of the reaction:
1 mole of manganese oxide reacts with 3 moles of hydrochloric acid.
So, 0.184 moles of manganese oxide will react with =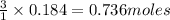 of hydrochloric acid
of hydrochloric acid
As, given amount of hydrochloric acid is more than the required amount. So, it is considered as an excess reagent.
Thus, manganese oxide is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
1 mole of manganese oxide reacts with 1 moles of chlorine.
So, 0.184 moles of manganese oxide will react with =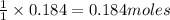 of chlorine.
of chlorine.
Now, calculating the mass of chlorine from equation 1, we get:
Molar mass of chlorine = 71 g/mol
Moles of chlorine = 0.186 moles
Putting values in equation 1, we get:
Hence, the mass of chlorine produced in the given reaction is 13.206 grams.
Ответ:
Step-by-step explanation: