maribelmartine8919
01.04.2021 •
Chemistry
How many liters of a 0.400 M HBr dilute solution can be made from a stock solution of 5.00 L of 8.00 M HBr solution?
Edit
View Insert Format Tools Table
Solved
Show answers
More tips
- H Health and Medicine Get Rid of Warts: Simple and Effective Ways...
- S Style and Beauty How to Choose the Perfect Hair Color?...
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Can someone me with geography connection plz....
- C Chemistry Nuclear fission occurs when an atomic nucleus splits into two or more smaller and less massive atomic nuclei. true false...
- C Chemistry In terms of potential energy, pe, which expression defines the heat of reaction for a chemical change?...
- C Chemistry What equation tells you how to calculate the number of neutrons in an atom? ?...
- C Chemistry Plzz me i m 90pts i m ! describe what would happen to both air temperature and soil temperature if cold weather were to pass through the area. what can you learn about...
- C Chemistry An electric energy coverts into what energy...
- C Chemistry The process responsible for upward movement of water in the water cycle is...
- C Chemistry Which energy conversion must occur in an operating electrolytic cell?...
- C Chemistry Ascientist gathered all of the available data based on the data, she tells us “what the data indicates” this is known as concluding predicting analyzing supposing...
- C Chemistry What is it called when water drops form something...

Ответ:
a. 6.8 moles of water
b. 41.6 moles of carbon dioxide
Explanation:
The balanced chemical equation for formation of water is:
According to stoichiometry :
1 mole of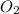 produces = 2 moles of
produces = 2 moles of 
Thus 3.4 moles of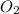 produce =
produce = of
of 
The balanced chemical equation for combustion of butane is:
According to stoichiometry :
1 mole of butane produces = 8 moles of
Thus 5.2 moles of butane produce = of
of 