How many minutes does it take light, at a speed of 3.00e8 m/s (meters per second) to travel a distance of 93 million miles from the Sun to the Earth?
Solved
Show answers
More tips
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- S Style and Beauty Unbelievable Hairstyles for Long Hair - 2021 Trends...
- H Health and Medicine When can it be said that a person has a normal pulse?...
Answers on questions: Chemistry
- B Business g A loan is taken to purchase experimental supplies for development of a personal device that can be used to obtain clean water from many common contaminated sources. The...
- M Mathematics Graph the relation. Is the relation a function? Why or why not? {(-5, 6), (-2, 3), (3, 2), (6, 4)} No; a range value has two domain values. Yes; there is only one range value...
- M Mathematics What are the values of m and n in kira’s equation...
- B Biology Which of the following statements describes a difference between how viruses reproduce and how cells reproduce? A. Viruses can reproduce sexually or asexually, whereas cells...

Ответ:
t = 8.27 minutes
Explanation:
We have,
Speed of light is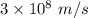
Distance of 93 million miles or
let t is time. So
or
t = 8.27 minutes
So, 8.27 minutes is taken by the light.
Ответ:
-
Mass of Hydrated KAl(SO₄)₂ = 2.0 g
Molar mass of anhydrous KAl(SO₄)₂ = 258.20 g/ mol
Mass of of anhydrous KAl(SO₄)₂ = mass of the 2nd heating = A
The mass of water released = mass of the Aluminum Cup + 2.0 grams of KAl(SO₄)₂ - mass of the 2nd heating
= H g
Moles of water released =
Moles of anhydrous KAl(SO₄)₂ =
Required ratio =