How many molecules are in 0.0124 mol of ch2o
Solved
Show answers
More tips
- S Society and Politics Why Did Medvedev Announce a New Privatization of State Property?...
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- C Chemistry SOMEONE HEL! Find the mass of a sample of water if its temperature dropped 24.8 °C when it lost 870 J of heat. Hint. Which formula are you going to use? Don t forget the...
- C Chemistry Calorimetry problem for chem please help no link answers will give brainliest and work would be appreciated...
- C Chemistry Help on this assessment...
- C Chemistry The barometric pressure in breckenridge colorado is (elevation 9600 frleet) is 580 mmhg. how many atmospheres is there...
- C Chemistry What is the most responsible for the present of groundwater in an area...
- C Chemistry Select the correct answer from each drop-down menu. what is the chemical name of this compound?...
- H History WHO WANNA HELP ME BECOME A CHRISTIAN...
- M Mathematics 2008 = NOOB and 426 = and...
- P Physics 1. Santa has the sleigh all loaded down with toys for all the good little girls and boys. There have been so many good children this year that the total mass for the Reindeer...
- C Computers and Technology What is the output for the following program? numA = 4 while numA 12: numA = numA + 3 print(numA) Output:...

Ответ:
There are 0.0747×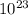 molecules in 0.0124 moles of
molecules in 0.0124 moles of 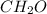 .
.
Explanation:
We know that 1 mole of any element is equal to Avagadro's number of molecules present in that element. So in this case, it is stated that 0.0124 moles of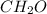 is present. Then the number of molecules will be obtained by multiplying the number of moles with Avagadro's number.
is present. Then the number of molecules will be obtained by multiplying the number of moles with Avagadro's number.
No. of molecules = No.of moles × Avagadro's number
No.of molecules = 0.0124×6.022×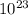 molecules =0.0747×
molecules =0.0747×
Thus, 0.0747×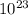 molecules of
molecules of 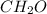 are present in 0.0124 moles of
are present in 0.0124 moles of 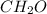 .
.
Ответ:
The horizontal distance represents wavelength λ. The time represents the period of the wave T.
Wave speed is given by v= λ/T
Hence, those are the only parameters needed.
Note: the vertical distance is for calculating amplitude which is not used in calculating wave speed.