davidaagurto
09.04.2021 •
Chemistry
How many moles are AgNO, are produced from 40 moles of HNO, according to the
following reaction:
3Ag + 4HNO3 3AgNO, + 2H2O + NO
A.10 mol AgNO3
B.20 mol AgNO3
C.30 mol AgNO3
D.40 mol AgNO3
Solved
Show answers
More tips
- H Health and Medicine Tick Traps: How to Remove Them Safely and Effectively...
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
- A Animals and plants Want a Perfect Lawn? Learn How to Plant Grass the Right Way...
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- C Chemistry Which of the following is an example of violating aufbau principle in filling electronic orbitals...
- C Chemistry What is the [H+] of a HCl solution that has a pH of 3.0?...
- C Chemistry The outermost electrons are theelectrons...
- C Chemistry Can you tell me about atoms...
- C Chemistry Help please help thank you...
- C Chemistry I visit the Grand Canyon I go to arizona.(once,whenever,wherever)...
- C Chemistry A gas has a pressure of 20 atm and a temperature of 400K. If I raise the pressure to 30 atm, what is the new temperature of the gas? A. List the VALUES of each the variables...
- C Chemistry How many moles are there in 6.06 X 1025 molecules Of H20?...
- C Chemistry The net ionic eq for Br ^+2 changes onto BrCrO4 in the presence of A acid B base C no reagent D none...
- C Chemistry The surface meaning of a word is that word s? ...

Ответ:
It is true about the type of reaction that it is a single replacement reaction, and the cations in the two ionic compounds are different.
Explanation:
When one element in a compound is replaced by another element in a chemical reaction then it is called a single replacement reaction.
For example,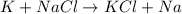
Here, potassium metal is replacing the sodium metal in the sodium chloride compound.
As metals become cation by losing an electron in a chemical reaction.
Thus, we can conclude that it is true about the type of reaction that it is a single replacement reaction, and the cations in the two ionic compounds are different.