penelopymorales
25.06.2019 •
Chemistry
How many moles are in 17.0 grams of h2o2? 0.284 mol h2o2 0.385 mol h2o2 0.500 mol h2o2 0.730 mol h2o2
Solved
Show answers
More tips
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry Convert 392 K into Celsius. Your answer should have three significant figures (round your answer to the nearest whole number). Use −273∘C for absolute zero....
- C Chemistry Write lewis structural formulas for the following molecules: nh3 so2 ch3oh hno2 n2 ch2o...
- C Chemistry Why is it not possible to just consume only pure atp and thus eliminate some steps in metabolism?...
- C Chemistry Need help with a chemistry question...
- H History The method of using reason to support Christian beliefs was known as...
- H History Can someone win the Electoral College without winning the popular vote?...
- M Mathematics The volume of a rectangular solid can be written as V=LWH. A Box of cereal has a width of 3inches. It s height is 4 inches longer tha it s length. If the volume of the...
- M Mathematics Help me please! Math homework. I’m in 7th grade, and I lost all my brain cells...
- M Mathematics 3x+2y=24, -x+2y=16 solve the system of linear equations by elimination....
- E English Which questions best demonstrate how to objectively evaluate an essay for interesting and unique presentation? Select two options....

Ответ:
Option (c) is the correct answer.
Explanation:
Number of moles is defined as given mass of substance divided by molar mass.
Mathematically, No. of moles =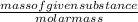
Molar mass of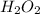 is 34.015 g/mol.
is 34.015 g/mol.
Therefore, calculate number of moles of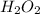 as follows.
as follows.
No. of moles =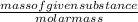
=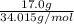
= 0.4997 mol
or, = 0.500 mol (approx)
Thus, we can conclude that 0.500 mol is present in 17.0 grams of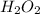 .
.
Ответ:
explanation:
if u put 3.74 that means there is three of something if u put .374 that means there is less than 1 of somethin if at makes sense.