How many moles of benzoic acid are present? How many moles of sodium bicarbonate are contained in 1 ml of 10% aqueous solution? (A 10% solution has 1 gram of solute in 9 mL of solvent.) Is the amount of sodium bicrbonate sufficient to react with benzoic acid.
Solved
Show answers
More tips
- H Health and Medicine What to Take with You to the Maternity Hospital?...
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- M Mathematics What is the value of x in the solution to the following system of linear equations?...
- C Chemistry Preguntas sobre el mundo natural. Plantear una hipótesis o posible respuesta a esa pregunta....
- M Mathematics 30 out of 120 students surveyed in JHS 185 participate in extracurricular activities. Nason is trying to create a simulation model for how many students participate...
- E Engineering A wet cooling tower is to cool 60 kg/s of water from 40 to 33°C. Atmospheric air enters the tower at 1 atm with dry-and wet-bulb temperatures of 22 and 16°C, respectively,...

Ответ:
Complete Question
In experiment 1, how many moles of benzoic acid are present? How many moles of sodium bicarbonate are contained in 1 ml of 10% aqueous solution? (A 10% solution has 1 gram of solute in 9 mL of solvent.) Is the amount of sodium bicarbonate sufficient to react with benzoic acid. The amount of Benzoic Acid in ex. 1 is 0.060 grams
No of moles of benzoic acid present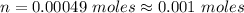
Number of moles of sodium bicarbonate present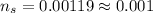
Yes the is sufficient sodium bicarbonate to react with Benzoic acid
Explanation:
From the question we are told that
The mass of Benzoic acid is
The number of moles of Benzoic acid is
where is the molar mass of Benzoic acid which is a constant with values
is the molar mass of Benzoic acid which is a constant with values
So
the density of the solution of sodium bicarbonate is
The mass of sodium bicarbonate is
The molar mass of sodium bicarbonate is a constant with value
The number of moles of sodium bicarbonate is
So the number of moles of Benzoic acid and sodium bicarbonate that react are approximately in a 1:1 ratio so we can conclude that there is sufficient sodium bicarbonate to react Benzoic acid
Ответ:
Step-by-step explanation:
Polynomials are:
consisting of indeterminates and coefficients, that involves only the operations of addition, subtraction, multiplication, and non-negative integer exponentiation of variables.According to definition of polynomials we can state:
Numbers 2, 5 and 6 are polynomialsNumbers 1, 3 and 4 are not polynomials as they have radicals, negative integer exponents and involve division operation.