exoticbunnylover123
28.08.2019 •
Chemistry
How many moles of chloride ions are in 0.2550 g of aluminum chloride using alcl3 's molar mass of 133.34 g / mol: ?
Solved
Show answers
More tips
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Any help would be good thanks...
- C Chemistry What would the molarity of a solution be if you had 80 grams NaOH that was added to 200 milliliters of water?...
- C Chemistry Which subatomic particles have approximately the same mass? protons and electrons neutrons and electrons protons and neutrons electrons and atoms...
- C Chemistry If vegetable oil is made out of vegetables and olive oil is made out of olives is baby oil made from baby’s...
- C Chemistry How many O atoms are in 7.39 g CO2?...
- C Chemistry A very thin-walled spherical storage tank has an outer diameter of 26 cm. The inner surface temperature of the tank is 413K. The spherical tank was covered with three insulation...
- C Chemistry Amixture of 50 wt% methane, 35 wt% ethane, and 15 wt% propane. determeine the mole fraction of methane. (can use a basis of 100kg) b) what is the average molecular weight of the...
- C Chemistry Calculate the internal energy and enthalpy variations for a heating isobaric process of ethanol from 150 ° f to 240 ° f, considering that cp is a function of the temperature....
- C Chemistry Adrilling mud is a slurry pumped into oil wells being drilled. the mud has several functions. it floats rock cuttings to the top of the well where they can easily be removed, lubricates...
- C Chemistry Many calculations require an absolute temperature, either kelvin or rankine as opposed to fahrenheit or celsius. as fall approaches we would like to know-is 400 r sweater weather?...

Ответ:
Explanation:
It is known that number of moles is the amount of mass present in a given molar mass of the substance.
Mathematically, No. of moles =
We are given that mass of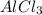 is 0.255 g and molar mass of
is 0.255 g and molar mass of 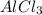 is 133.34 g/mol.
is 133.34 g/mol.
Therefore, number of moles of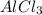 is calculated as follows.
is calculated as follows.
No. of moles =
=
= mol
mol
Since, there are three chlorine atoms present in one mole of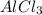 . Therefore, moles of chlorine ions present in given 0.255 g will be calculated as follows.
. Therefore, moles of chlorine ions present in given 0.255 g will be calculated as follows.
=
Thus, we can conclude that chloride ions are in 0.2550 g of aluminum chloride using
chloride ions are in 0.2550 g of aluminum chloride using 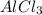 's molar mass of 133.34 g/mol.
's molar mass of 133.34 g/mol.
Ответ:
Ответ: