alayjared12374
17.07.2019 •
Chemistry
How many moles of gas are present in a 1.07 l sample at 1.56 atm and a temperature of 22.0oc?
Solved
Show answers
More tips
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- M Mathematics Which of the following is not considered a piecewise function?...
- P Physics Calculate the volume of a ball having radius of 8 cm...
- B Business 2 Nina is a social media manager for a small indoor trampoline business. Nina s job is important because she spreads the word about their special events. What quality...
- M Mathematics What is the volume of the prism the units are 7m 24m and 21m...
- M Mathematics When y=75,x=1 over 2 what is the value of y when x is 2 and 1/4...

Ответ:
V(gas) = 1.07 L; volume.
T = 22.0°C = 295.15 K; temperature of gas.
p = 1.56 atm; pressure of gas.
R = 0.08206 L·atm/mol·K; gas constant.
Ideal gas law: p·V = n·R·T.
n = p·V / R·T.
n(gas) = 1.56 atm · 1.07 L / 0.08206 L·atm/mol·K · 295.15 K.
n(gas) = 0.069 mol; amount of substance.
Ответ:
Approximately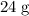 (at most.)
(at most.)
Explanation:
Aluminum reacts with bromine
reacts with bromine  at a
at a  ratio:
ratio:
Look up the relative atomic mass of and
and  . From a modern periodic table:
. From a modern periodic table:
Calculate the formula mass of the reactants and of the product:
Calculate the quantity (in number of moles of formula units) of each reactant:
Assume that is the limiting reactant. From the coefficients:
is the limiting reactant. From the coefficients:
Based on the assumption that is the limiting reactant:
is the limiting reactant:
In other words, if is the limiting reactant (meaning that
is the limiting reactant (meaning that  is in excess,) then approximately
is in excess,) then approximately 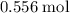 of
of  will be produced.
will be produced.
On the other hand, assume that is the limiting reactant. Similarly, from the coefficients:
is the limiting reactant. Similarly, from the coefficients:
Based on the assumption that is the limiting reactant:
is the limiting reactant:
Compare the value based on the two assumptions. Only the smallest value,
value based on the two assumptions. Only the smallest value,  (under the assumption that
(under the assumption that  is the limiting reactant,) would resemble the theoretical yield. The reason is that
is the limiting reactant,) would resemble the theoretical yield. The reason is that  would run out before all that
would run out before all that  of
of  was converted to
was converted to  .
.
Apply the formula mass of to find the mass of that (approximately)
to find the mass of that (approximately)  of
of  formula units:
formula units: