erikamaldonado661
03.04.2021 •
Chemistry
how much alum product would be lost to the crystallization solution if you had 42.5 ml of solution after filtration and the solubility of alum is approximately 2.63 g alum in 100 ml of 0 degrees celcius acidic water
Solved
Show answers
More tips
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
Answers on questions: Chemistry
- C Chemistry When the ideal gas law constant 0.0821 is used, what unit of pressure should be used in the ideal gas law calculation?...
- C Chemistry Ineed with these 2 chem questions. i’m stuck! this is college level chem...
- C Chemistry Ineed with these 2 questions. i’m stuck!...
- C Chemistry Trigonal planar vs. trigonal pryramidal difference? how to determine whether a bond is one or the other...
- C Chemistry How much work will it take to lift a 4-kg baby 1 meter into her crib?...
- C Chemistry What is corrosion? what is corrosion of iron called?...
- C Chemistry Toasting bread what type of heat transfer...
- C Chemistry Did i get all of those questions right? if is not tell me why that is wrong?...
- C Chemistry What is civic expression?...
- C Chemistry The molar mass of sodium hydroxide is 39.997 g per mole. If 98 g of NaOH are dissolved into 2.2 L of solution, what is the molarity?...

Ответ:
Total amount of alum lost = 0.5122 grams
Explanation:
Let the total volume of the solution be 100 mL
In 100 mL of solution, there is 2.63 gram of alum.
Out of this 100 mL solution, 42.5 mL is remaining.
Amount of alum in 42.5 mL solution is
Now the amount of alum lost is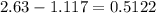 grams
grams
Ответ:
herpes
Explanation: