southsan2021
18.10.2019 •
Chemistry
Hurry will mark brainliest
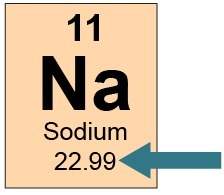
this is how sodium appears in the periodic table.
the arrow is pointing to sodium’s
number of neutrons.
number of valence electrons.
combined number of neutrons and protons.
combined number of electrons and protons.
Solved
Show answers
More tips
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
Answers on questions: Chemistry
- M Mathematics What is the area of the object above? (Picture Attached :D)...
- B Business All of the five elements of manufacturing contribute to the cost of production. which one of the following is not one of the 5ms? a. methods b. mandates c. measurement d. machinery...
- M Mathematics A15 foot rope stretches diagonally from the ground to the top of a tent 9 feet off the ground. what angle does the rope form with the ground?...
- M Mathematics Find the value of x....

Ответ:
12 Neutrons
1 Valence Electron
23 ions [11 Protons and 12 Neutrons]
22 ions [11 Electrons and Protons]
Explanation:
To find the number of neutrons in an element, you simply take its atomic number and deduct that from the atomic mass [round if necessary]. There is one valence electron because according to the Periodic Table of Elements, the first three energy levels can fit 2 - 8 electrons:
1st Energy Level → 2 Electrons
2nd Energy Level → 8 Electrons
3rd Energy Level → 8 Electrons
4th Energy Level → 18 Electrons
5th Energy Level → 18 Electrons
6th Energy Level [Lanthanide Series] → 32 Electrons
7th Energy Level [Actinide Series] → 32 Electrons
So, as you can see, 2 + 8 gives you 10. So from there, we already filled up 2 energy levels. Now going to the third one will leave us with only one electron left over. That is where that valence electron is.
** I encourage you to write this down in your Chemistry notebook somewhere, so you can keep this stored in your memory at all times. There are some pointers in the answer as well. Copy that.
I am joyous to assist you anytime.
Ответ:
(Apex Learning Answer) Study how cholesterol is broken down in the body.
Explanation: