wcraig1998
10.03.2021 •
Chemistry
I need help, please answer
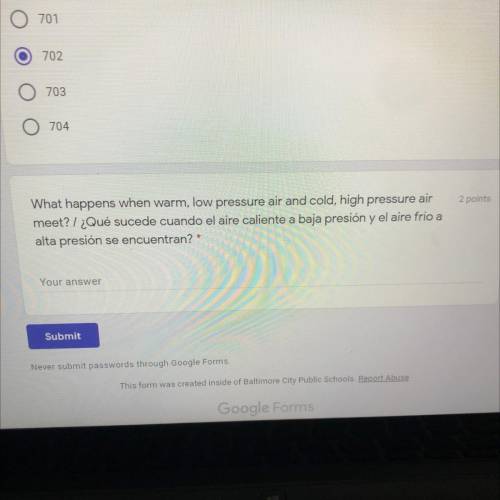
Solved
Show answers
More tips
- A Art and Culture How to Learn to Sing? A Complete Guide for Beginners...
- H Health and Medicine How to Get Rid of Dandruff?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
Answers on questions: Chemistry
- C Chemistry Chooses all of the terms that refers to an ethnic...
- C Chemistry Copper(1) oxide will react with oxygen gas to produce copper(ll) oxide. If 95.3 g of copper(1) oxide is reacted, how many grams of oxygen will be needed?...
- C Chemistry If 5.00 g CuCl2 reacted, how many moles of copper could form?...
- C Chemistry Identify each charge of the subatomic particles ? Help me please ASAP...
- C Chemistry A structure that can lessen erosion is...
- C Chemistry Increasing which variable would decrease the pressure of a contained gas?...
- C Chemistry Warm air lifted over a moving cold air mass will produce a front...
- C Chemistry All of the following are possible sources of error in a scientific investigation except for...
- C Chemistry A solution is prepared initially containing 0.050 M Pb(NO3)2 and 0.20 M NaF. What will be the concentration of all ions after precipitation of PbF2 occurs? Answers: na=.20M, no3^-=...
- C Chemistry What is the relationship between earth and the property of water as a universal solvent?...

Ответ:
it explodes
Explanation:
Ответ:
Mrs girl I'm sorry I don't know
I just need the points
Ответ:
C. 588 nm
Explanation:
Given parameters:
Energy of the photon = 3.38 x 10⁻¹⁹J
Unknown:
Wavelength of the photon = ?
Solution:
The energy of a photon can be expressed as;
E =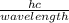
hc = E x wavelength
Wavelength =
h is the Planck's constant = 6.63 x 10⁻³⁴m²kg/s
c is the speed of light = 3 x 10⁸m/s
E is the energy
Wavelength = = 5.89 x 10⁻⁷m
= 5.89 x 10⁻⁷m