quinteroalex2001
18.05.2021 •
Chemistry
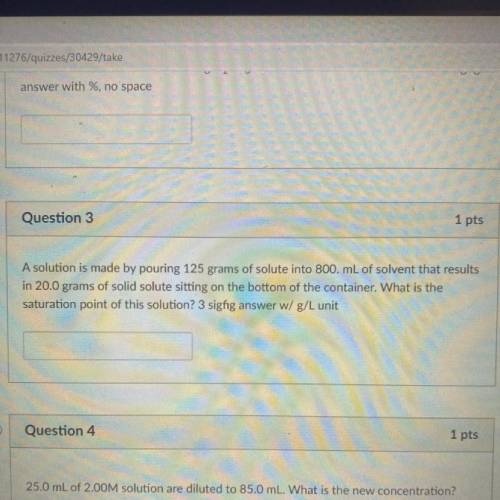
I need help with #3 please and Thank you so so much
Solved
Show answers
More tips
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- G Goods and services What Are the Most Popular Services?...
- O Other What is the oldest joke ever told?...
- L Legal consultation How to Properly Inherit: Tips and Recommendations...
- C Computers and Internet Boost your processor performance with these easy tips...
- S Sport How does Bodyflex work: what is it and how does it work?...
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- S Style and Beauty How to Properly Apply Eye Makeup: Tips from a Professional Makeup Artist...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
Answers on questions: Chemistry
- C Chemistry A student performs an experiment three times. The results are 4.52 g/mL, 4.54 g/mL, and 4.39 g/mL. Which of the following best describes these results with only the information...
- C Chemistry 6 A student is making a solution of potassium permanganate(KMnO.). They mix 23.7 grams of potassium permanganate into 750.0 mL of water. What is the molarity of the potassium...
- C Chemistry What is amount of energy released by the reaction? O 5 10 15 020 025 30 35 040 45 50...
- C Chemistry How much of a 3.75 M KI solution would you need to prepare 252.5 mL of a 0.780 M KI solution?...
- C Chemistry Identify the compound with the highest vapor pressure....
- C Chemistry If the [H3O+] = 7.5x10^-9 M, find the poH two ways....
- C Chemistry Acetylene gas, C2H2, is produced as a result of the following reaction: CaC2 + 2H2O C2H2 + Ca(OH)2. (a) If 32 grams of CaC2 are consumed in this reaction, how many moles...
- C Chemistry What does a man s home is his castle mean...
- C Chemistry Need question in the picture...
- C Chemistry Liquids and solids are known as states of matter...

Ответ:
Explanation is in the answer
Explanation:
The pH of the buffer solution does not change appreciably because the strong acid (free H⁺) reacts with conjugate base of buffer producing more weak acid. pH formula of buffers is (Henderson-Hasselbalch formula):
pH = pKa + log ( [A⁻] / [HA] )
The addition of strong acid decreases [A⁻] increasing [HA]. pH change just in the log of the ratio of [A⁻] with [HA], that is a real little effect over pH of the buffer solution.