itcelmairani
01.06.2021 •
Chemistry
I need help with this question please help
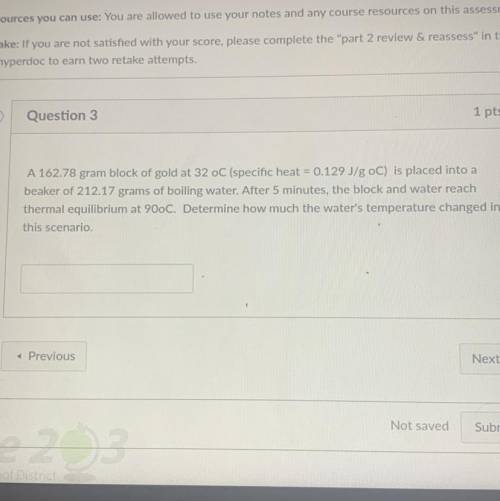
Solved
Show answers
More tips
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- E English Which character in the story do you most admire and why? from the book killing Mr. Griffin write an essay in this topic pls help...
- S Social Studies All countries in Western Europe have a level of development that is considered ....
- P Physics Freud was a influential and controversial psychologist who believed that psychology should study the unconscious mind. Describe the two books Freud published in the...
- M Mathematics What is the solution set of the quadratic inequality 4(x + 2)2 0?...

Ответ:
deez
Explanation:
Ответ:
The time needed for the reaction will increase.
Explanation:
As we know from kinetics of chemical reaction that the rate of the reaction (except zero order reactions) depends on the concentration of the reactants.Rate of chemical reaction α [reactants].If we suppose here that the reaction is a second order reaction which means that the reaction depends on the concentration of both H₂SO₄ and thiosulfate.Rate of the reaction = k[H₂SO₄][thiosulfate].
where, k is the rate constant of the reaction.Here, the concentration of H₂SO₄ is constant, since we use a constant a mount of it.The concentration of thiosulfate decreased from flask 1 to flask 5 as the amount of added thiosulfate decreased.Rate α [thiosulfate]
As the concentration of the thiosulfate decreased, the rate of the reaction will decrease.The rate of the reaction is the change in the concentration of the reactants per time.Rate of the reaction = - Δ[thiosulfate]/Δt.
As we mentioned that the concentration of thiosulfate decreased, since the rate of the reaction will decrease.The rate of the reaction is inversely proportional to time (rate α 1/t).The time needed for the reaction will increase from flask 1 to flask 5.
Rights reserved to their respective writers®